Ciliogenesis associated kinase 1: targets and functions in various organ systems
- PMID: 31506943
- PMCID: PMC6848764
- DOI: 10.1002/1873-3468.13600
Ciliogenesis associated kinase 1: targets and functions in various organ systems
Abstract
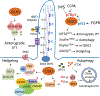
Ciliogenesis associated kinase 1 (CILK1) was previously known as intestinal cell kinase because it was cloned from that origin. However, CILK1 is now recognized as a widely expressed and highly conserved serine/threonine protein kinase. Mutations in the human CILK1 gene have been associated with ciliopathies, a group of human genetic disorders with defects in the primary cilium. In mice, both Cilk1 knock-out and Cilk1 knock-in mutations have recapitulated human ciliopathies. Thus, CILK1 has a fundamental role in the function of the cilium. Several candidate substrates have been proposed for CILK1 and the challenge is to relate these to the mutant phenotypes. In this review, we summarize what is known about CILK1 functions and targets, and discuss gaps in current knowledge that motivate further experimentation to fully understand the role of CILK1 in organ development in humans.
Keywords: autophagy; ciliogenesis; ciliopathy; epilepsy; hedgehog signalling; intraflagellar transport; kinesin family member 3A; primary cilia; stereocilia.
© 2019 Federation of European Biochemical Societies.
Figures
Similar articles
-
Phosphosite T674A mutation in kinesin family member 3A fails to reproduce tissue and ciliary defects characteristic of CILK1 loss of function.Dev Dyn. 2021 Feb;250(2):263-273. doi: 10.1002/dvdy.252. Epub 2020 Oct 7. Dev Dyn. 2021. PMID: 32935890 Free PMC article.
-
Functional Alterations in Ciliogenesis-Associated Kinase 1 (CILK1) that Result from Mutations Linked to Juvenile Myoclonic Epilepsy.Cells. 2020 Mar 12;9(3):694. doi: 10.3390/cells9030694. Cells. 2020. PMID: 32178256 Free PMC article.
-
Modulation of Primary Cilia by Alvocidib Inhibition of CILK1.Int J Mol Sci. 2022 Jul 23;23(15):8121. doi: 10.3390/ijms23158121. Int J Mol Sci. 2022. PMID: 35897693 Free PMC article.
-
Genes and molecular pathways underpinning ciliopathies.Nat Rev Mol Cell Biol. 2017 Sep;18(9):533-547. doi: 10.1038/nrm.2017.60. Epub 2017 Jul 12. Nat Rev Mol Cell Biol. 2017. PMID: 28698599 Free PMC article. Review.
-
CPLANE Complex and Ciliopathies.Biomolecules. 2022 Jun 17;12(6):847. doi: 10.3390/biom12060847. Biomolecules. 2022. PMID: 35740972 Free PMC article. Review.
Cited by
-
Appearing and disappearing acts of cilia.J Biosci. 2023;48(1):8. doi: 10.1007/s12038-023-00326-6. J Biosci. 2023. PMID: 36924208 Free PMC article. Review.
-
Prostaglandin Analogs and Eupatilin as Treatments for Nephronophthisis.Kidney Int Rep. 2025 May 2;10(8):2821-2835. doi: 10.1016/j.ekir.2025.04.060. eCollection 2025 Aug. Kidney Int Rep. 2025. PMID: 40814642 Free PMC article.
-
Kinase-dependent regulation of ciliary protein transport and its implications for therapy.Front Mol Biosci. 2025 Jun 25;12:1638737. doi: 10.3389/fmolb.2025.1638737. eCollection 2025. Front Mol Biosci. 2025. PMID: 40636449 Free PMC article. Review.
-
Ciliary Hedgehog signaling patterns the digestive system to generate mechanical forces driving elongation.Nat Commun. 2021 Dec 10;12(1):7186. doi: 10.1038/s41467-021-27319-z. Nat Commun. 2021. PMID: 34893605 Free PMC article.
-
Phosphosite T674A mutation in kinesin family member 3A fails to reproduce tissue and ciliary defects characteristic of CILK1 loss of function.Dev Dyn. 2021 Feb;250(2):263-273. doi: 10.1002/dvdy.252. Epub 2020 Oct 7. Dev Dyn. 2021. PMID: 32935890 Free PMC article.
References
-
- Bladt F, and Birchmeier C (1993) Characterization and expression analysis of the murine rck gene: a protein kinase with a potential function in sensory cells. Differentiation 53, 115–122 - PubMed
-
- Abe S, Yagi T, Ishiyama S, Hiroe M, Marumo F, and Ikawa Y (1995) Molecular cloning of a novel serine/threonine kinase, MRK, possibly involved in cardiac development. Oncogene 11, 2187–2195 - PubMed
-
- Togawa K, Yan YX, Inomoto T, Slaugenhaupt S, and Rustgi AK (2000) Intestinal cell kinase (ICK) localizes to the crypt region and requires a dual phosphorylation site found in map kinases. J Cell Physiol 183, 129–139 - PubMed
-
- Miyata Y, Akashi M, and Nishida E (1999) Molecular cloning and characterization of a novel member of the MAP kinase superfamily. Genes Cells 4, 299–309 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

