Plasmonic Photothermal Nanoparticles for Biomedical Applications
- PMID: 31508273
- PMCID: PMC6724476
- DOI: 10.1002/advs.201900471
Plasmonic Photothermal Nanoparticles for Biomedical Applications
Abstract
Recent advances of plasmonic nanoparticles include fascinating developments in the fields of energy, catalyst chemistry, optics, biotechnology, and medicine. The plasmonic photothermal properties of metallic nanoparticles are of enormous interest in biomedical fields because of their strong and tunable optical response and the capability to manipulate the photothermal effect by an external light source. To date, most biomedical applications using photothermal nanoparticles have focused on photothermal therapy; however, to fully realize the potential of these particles for clinical and other applications, the fundamental properties of photothermal nanoparticles need to be better understood and controlled, and the photothermal effect-based diagnosis, treatment, and theranostics should be thoroughly explored. This Progress Report summarizes recent advances in the understanding and applications of plasmonic photothermal nanoparticles, particularly for sensing, imaging, therapy, and drug delivery, and discusses the future directions of these fields.
Keywords: metal nanoparticles; phothermal effect; photothermal therapy; plasmonic nanoparticles; theranostics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

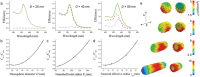
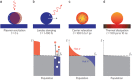
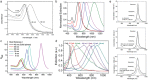




Similar articles
-
Plasmonic nanoparticle-based surface-enhanced Raman spectroscopy-guided photothermal therapy: emerging cancer theranostics.Nanomedicine (Lond). 2023 Mar;18(6):555-576. doi: 10.2217/nnm-2023-0010. Epub 2023 May 18. Nanomedicine (Lond). 2023. PMID: 37199287 Review.
-
Magnetic-Plasmonic Core-Shell Nanoparticles: Properties, Synthesis and Applications for Cancer Detection and Treatment.Nanomaterials (Basel). 2025 Feb 10;15(4):264. doi: 10.3390/nano15040264. Nanomaterials (Basel). 2025. PMID: 39997827 Free PMC article. Review.
-
Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine.Acc Chem Res. 2008 Dec;41(12):1578-86. doi: 10.1021/ar7002804. Acc Chem Res. 2008. PMID: 18447366
-
Coiled-coil peptide-based assembly of a plasmonic core-satellite polymer-metal nanocomposite as an efficient photothermal agent for drug delivery applications.J Colloid Interface Sci. 2023 Jul;641:929-941. doi: 10.1016/j.jcis.2023.03.085. Epub 2023 Mar 16. J Colloid Interface Sci. 2023. PMID: 36989819
-
Plasmonic nanoparticle-generated photothermal bubbles and their biomedical applications.Nanomedicine (Lond). 2009 Oct;4(7):813-45. doi: 10.2217/nnm.09.59. Nanomedicine (Lond). 2009. PMID: 19839816 Free PMC article.
Cited by
-
Remotely Activated Nanoparticles for Anticancer Therapy.Nanomicro Lett. 2020 Oct 27;13(1):11. doi: 10.1007/s40820-020-00537-8. Nanomicro Lett. 2020. PMID: 34138198 Free PMC article. Review.
-
Antibacterial Evaluation of Zirconia Coated with Plasma-Based Graphene Oxide with Photothermal Properties.Int J Mol Sci. 2023 May 17;24(10):8888. doi: 10.3390/ijms24108888. Int J Mol Sci. 2023. PMID: 37240234 Free PMC article.
-
Nanoparticles for Interrogation of Cell Signaling.Annu Rev Anal Chem (Palo Alto Calif). 2023 Jun 14;16(1):333-351. doi: 10.1146/annurev-anchem-092822-085852. Annu Rev Anal Chem (Palo Alto Calif). 2023. PMID: 37314874 Free PMC article. Review.
-
Magnetite Nanoparticles and Spheres for Chemo- and Photothermal Therapy of Hepatocellular Carcinoma in vitro.Int J Nanomedicine. 2020 Oct 14;15:7923-7936. doi: 10.2147/IJN.S257142. eCollection 2020. Int J Nanomedicine. 2020. PMID: 33116509 Free PMC article.
-
Nanobiosensors for the Detection of Novel Coronavirus 2019-nCoV and Other Pandemic/Epidemic Respiratory Viruses: A Review.Sensors (Basel). 2020 Nov 18;20(22):6591. doi: 10.3390/s20226591. Sensors (Basel). 2020. PMID: 33218097 Free PMC article. Review.
References
-
- Puchkova A., Vietz C., Pibiri E., Wünsch B., Sanz Paz M., Acuna G. P., Tinnefeld P., Nano Lett. 2015, 15, 8354. - PubMed
-
- Ament I., Prasad J., Henkel A., Schmachtel S., Sönnichsen C., Nano Lett. 2012, 12, 1092. - PubMed
-
- Cole L. E., Ross R. D., Tilley J. M., Vargo‐Gogola T., Roeder R. K., Nanomedicine 2015, 10, 321. - PubMed
-
- Link S., El‐Sayed M. A., Int. Rev. Phys. Chem. 2000, 19, 409.
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
