Pristimerin attenuates cell proliferation of uveal melanoma cells by inhibiting insulin-like growth factor-1 receptor and its downstream pathways
- PMID: 31508890
- PMCID: PMC6815816
- DOI: 10.1111/jcmm.14623
Pristimerin attenuates cell proliferation of uveal melanoma cells by inhibiting insulin-like growth factor-1 receptor and its downstream pathways
Abstract
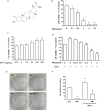
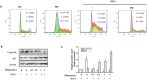
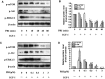
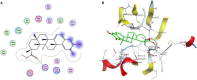
Uveal melanoma (UM) has a high mortality rate due to liver metastasis. The insulin-like growth factor-1 receptor (IGF-1R) is highly expressed in UM and has been shown to be associated with hepatic metastases. Targeting IGF signalling may be considered as a promising approach to inhibit the process of metastatic UM cells. Pristimerin (PRI) has been demonstrated to inhibit the growth of several cancer cells, but its role and underlying mechanisms in the IGF-1-induced UM cell proliferation are largely unknown. The present study examined the anti-proliferative effect of PRI on UM cells and its possible role in IGF-1R signalling transduction. MTT and clonogenic assays were used to determine the role of PRI in the proliferation of UM cells. Flow cytometry was performed to detect the effect of PRI on the cell cycle distribution of UM cells. Western blotting was carried out to assess the effects of PRI and IGF-1 on the IGF-1R phosphorylation and its downstream targets. The results indicated that IGF-1 promoted the UM cell proliferation and improved the level of IGF-1R phosphorylation, whereas PRI attenuated the effect of IGF-1. Interestingly, PRI could not only induce the G1 phase accumulation and reduce the G2 phase induced by IGF-1, but also could stimulate the expression of p21 and inhibit the expression of cyclin D1. Besides, PRI could attenuate the phosphorylations of Akt, mTOR and ERK1/2 induced by IGF-1. Furthermore, the molecular docking study also demonstrated that PRI had potential inhibitory effects on IGF-1R. Taken together, these results indicated that PRI could inhibit the proliferation of UM cells through down-regulation of phosphorylated IGF-1R and its downstream signalling.
Keywords: Akt; ERK1/2; IGF-1; mTOR; pristimerin; uveal melanoma.
© 2019 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Pristimerin-induced uveal melanoma cell death via inhibiting PI3K/Akt/FoxO3a signalling pathway.J Cell Mol Med. 2020 Jun;24(11):6208-6219. doi: 10.1111/jcmm.15249. Epub 2020 Apr 29. J Cell Mol Med. 2020. PMID: 32347651 Free PMC article.
-
Elucidating the role of the FoxO3a transcription factor in the IGF-1-induced migration and invasion of uveal melanoma cancer cells.Biomed Pharmacother. 2016 Dec;84:1538-1550. doi: 10.1016/j.biopha.2016.11.027. Epub 2016 Nov 20. Biomed Pharmacother. 2016. PMID: 27881235
-
Expression of insulin-like growth factor-1 receptor in metastatic uveal melanoma and implications for potential autocrine and paracrine tumor cell growth.Pigment Cell Melanoma Res. 2014 Mar;27(2):297-308. doi: 10.1111/pcmr.12206. Epub 2014 Jan 6. Pigment Cell Melanoma Res. 2014. PMID: 24354797
-
Insulin-like growth factor system and sporadic malignant melanoma.Am J Pathol. 2011 Jan;178(1):26-31. doi: 10.1016/j.ajpath.2010.11.004. Epub 2010 Dec 23. Am J Pathol. 2011. PMID: 21224039 Free PMC article. Review.
-
The application of natural compounds in uveal melanoma drug discovery.J Pharm Pharmacol. 2022 May 20;74(5):660-680. doi: 10.1093/jpp/rgac009. J Pharm Pharmacol. 2022. PMID: 35532546 Review.
Cited by
-
Pristimerin Exerts Pharmacological Effects Through Multiple Signaling Pathways: A Comprehensive Review.Drug Des Devel Ther. 2024 May 18;18:1673-1694. doi: 10.2147/DDDT.S460093. eCollection 2024. Drug Des Devel Ther. 2024. PMID: 38779590 Free PMC article. Review.
-
Transcriptome Profiling Reveals New Insights into the Immune Microenvironment and Upregulation of Novel Biomarkers in Metastatic Uveal Melanoma.Cancers (Basel). 2020 Sep 30;12(10):2832. doi: 10.3390/cancers12102832. Cancers (Basel). 2020. PMID: 33008022 Free PMC article.
-
Novel Prognostic Immunohistochemical Markers in Uveal Melanoma-Literature Review.Cancers (Basel). 2021 Aug 10;13(16):4031. doi: 10.3390/cancers13164031. Cancers (Basel). 2021. PMID: 34439182 Free PMC article. Review.
-
Skin anomalies in acromegalic patients (Review of the practical aspects).Exp Ther Med. 2021 Nov;22(5):1330. doi: 10.3892/etm.2021.10765. Epub 2021 Sep 20. Exp Ther Med. 2021. PMID: 34630684 Free PMC article. Review.
-
Cellular and Molecular Mechanisms of Pristimerin in Cancer Therapy: Recent Advances.Front Oncol. 2021 May 7;11:671548. doi: 10.3389/fonc.2021.671548. eCollection 2021. Front Oncol. 2021. PMID: 34026649 Free PMC article. Review.
References
-
- Augsburger JJ, Correa ZM, Shaikh AH. Effectiveness of treatments for metastatic uveal melanoma. Am J Ophthalmol. 2009;148:119‐127. - PubMed
-
- Economou MA, All‐Ericsson C, Bykov V, et al. Receptors for the liver synthesized growth factors IGF‐1 and HGF/SF in uveal melanoma: intercorrelation and prognostic implications. Acta Ophthalmologica. 2008;86(thesis4):20‐25. - PubMed
-
- Tognon CE, Sorensen PH. Targeting the insulin‐like growth factor 1 receptor (IGF1R) signaling pathway for cancer therapy. Expert Opin Ther Targets. 2012;16:33‐48. - PubMed
-
- Karamouzis MV, Papavassiliou AG. Targeting insulin‐like growth factor in breast cancer therapeutics. Crit Rev Oncol Hematol. 2012;84:8‐17. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

