Genomic deletion of TLR2 induces aggravated white matter damage and deteriorated neurobehavioral functions in mouse models of Alzheimer's disease
- PMID: 31509519
- PMCID: PMC6756907
- DOI: 10.18632/aging.102260
Genomic deletion of TLR2 induces aggravated white matter damage and deteriorated neurobehavioral functions in mouse models of Alzheimer's disease
Abstract
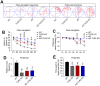
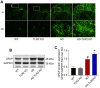
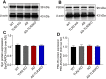
Toll-like receptor-2 (TLR2), a member of the TLR family, plays an important role in the initiation and regulation of immune/inflammation response, which is a critical mechanism underlying Alzheimer's disease (AD). To clarify the role of TLR2 in the pathological process of AD, in the present study, TLR2 knockout plus APPswe/PSEN1dE9 transgenic mice (AD-TLR2KO) were generated. Neurobehavioral tests and brain MRI scan were conducted on mice at the age of 12 months. Additionally, neuron loss was evaluated using NeuN staining. Amyloid β protein (Aβ), glial fibrillary acidic protein (GFAP), endogenous ligands for TLR2, and the activation of downstream signaling of TLR2 in mouse brains were detected by immunohistochemistry and Western blots. The results demonstrated that TLR2 deficit induced learning disabilities, decreased spontaneous activity, increased anxiety and depression, and led to white matter damage (WMD), brain atrophy, loss of neurons, and glial activation. Moreover, TLR2 deficit aggravated impaired neurobehavioral functions and WMD in AD mice, but did not affect the Aβ deposition in mouse brains. Our data indicate that the genomic deletion of TLR2 impairs neurobehavioral functions, induces WMD and brain atrophy, and increases the activation of astrocytes, which in turn aggravate the symptoms of AD through a non-Aβ mechanism.
Keywords: Alzheimer’s disease; MRI; TLR2; neurobehavioral function; white matter damage.
Conflict of interest statement
Figures







Similar articles
-
Inhibiting TLR2 activation attenuates amyloid accumulation and glial activation in a mouse model of Alzheimer's disease.Brain Behav Immun. 2016 Nov;58:191-200. doi: 10.1016/j.bbi.2016.07.143. Epub 2016 Jul 12. Brain Behav Immun. 2016. PMID: 27422717
-
MMP9 modulation improves specific neurobehavioral deficits in a mouse model of Alzheimer's disease.BMC Neurosci. 2021 May 25;22(1):39. doi: 10.1186/s12868-021-00643-2. BMC Neurosci. 2021. PMID: 34034683 Free PMC article.
-
Characterization of a 3xTg-AD mouse model of Alzheimer's disease with the senescence accelerated mouse prone 8 (SAMP8) background.Synapse. 2018 Apr;72(4). doi: 10.1002/syn.22025. Epub 2018 Feb 1. Synapse. 2018. PMID: 29341269
-
Selective disruption of TLR2-MyD88 interaction inhibits inflammation and attenuates Alzheimer's pathology.J Clin Invest. 2018 Oct 1;128(10):4297-4312. doi: 10.1172/JCI96209. Epub 2018 Jul 10. J Clin Invest. 2018. PMID: 29990310 Free PMC article.
-
Genetic Deletion of Tumor Necrosis Factor-α Attenuates Amyloid-β Production and Decreases Amyloid Plaque Formation and Glial Response in the 5XFAD Model of Alzheimer's Disease.J Alzheimers Dis. 2017;60(1):165-181. doi: 10.3233/JAD-170065. J Alzheimers Dis. 2017. PMID: 28826177
Cited by
-
Cellular Reprogramming and Its Potential Application in Alzheimer's Disease.Front Neurosci. 2022 Apr 7;16:884667. doi: 10.3389/fnins.2022.884667. eCollection 2022. Front Neurosci. 2022. PMID: 35464309 Free PMC article. Review.
-
Magnetic Resonance Imaging in Animal Models of Alzheimer's Disease Amyloidosis.Int J Mol Sci. 2021 Nov 25;22(23):12768. doi: 10.3390/ijms222312768. Int J Mol Sci. 2021. PMID: 34884573 Free PMC article. Review.
-
Impact of the SARS-CoV-2 Spike Protein on the Innate Immune System: A Review.Cureus. 2024 Mar 26;16(3):e57008. doi: 10.7759/cureus.57008. eCollection 2024 Mar. Cureus. 2024. PMID: 38549864 Free PMC article. Review.
-
Late-Stage Activation of Toll-like receptor 3 Alleviates Cognitive Impairment and Neuropathology in an Alzheimer's Disease Mouse Model.Mol Neurobiol. 2025 May 28. doi: 10.1007/s12035-025-05092-0. Online ahead of print. Mol Neurobiol. 2025. PMID: 40437286
-
From Immunity to Neurogenesis: Toll-like Receptors as Versatile Regulators in the Nervous System.Int J Mol Sci. 2024 May 24;25(11):5711. doi: 10.3390/ijms25115711. Int J Mol Sci. 2024. PMID: 38891900 Free PMC article. Review.
References
-
- Alzheimer’s Association. 2018 Alzheimer’s disease facts and figures. Alzheimers Dement. 2018; 14:367–429. 10.1016/j.jalz.2018.02.001 - DOI
-
- Wang C, Najm R, Xu Q, Jeong DE, Walker D, Balestra ME, Yoon SY, Yuan H, Li G, Miller ZA, Miller BL, Malloy MJ, Huang Y. Gain of toxic apolipoprotein E4 effects in human iPSC-derived neurons is ameliorated by a small-molecule structure corrector. Nat Med. 2018; 24:647–57. 10.1038/s41591-018-0004-z - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

