Neuronal Circuit Activity during Neonatal Hypoxic-Ischemic Seizures in Mice
- PMID: 31509619
- PMCID: PMC7025736
- DOI: 10.1002/ana.25601
Neuronal Circuit Activity during Neonatal Hypoxic-Ischemic Seizures in Mice
Abstract
Objective: To identify circuits active during neonatal hypoxic-ischemic (HI) seizures and seizure propagation using electroencephalography (EEG), behavior, and whole-brain neuronal activity mapping.
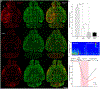
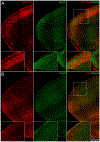
Methods: Mice were exposed to HI on postnatal day 10 using unilateral carotid ligation and global hypoxia. EEG and video were recorded for the duration of the experiment. Using immediate early gene reporter mice, active cells expressing cfos were permanently tagged with reporter protein tdTomato during a 90-minute window. After 1 week, allowing maximal expression of the reporter protein, whole brains were processed, lipid cleared, and imaged with confocal microscopy. Whole-brain reconstruction and analysis of active neurons (colocalized tdTomato/NeuN) were performed.
Results: HI resulted in seizure behaviors that were bilateral or unilateral tonic-clonic and nonconvulsive in this model. Mice exhibited characteristic EEG background patterns such as burst suppression and suppression. Neuronal activity mapping revealed bilateral motor cortex and unilateral, ischemic somatosensory cortex, lateral thalamus, and hippocampal circuit activation. Immunohistochemical analysis revealed regional differences in myelination, which coincide with these activity patterns. Astrocytes and blood vessel endothelial cells also expressed cfos during HI.
Interpretation: Using a combination of EEG, seizure semiology analysis, and whole-brain neuronal activity mapping, we suggest that this rodent model of neonatal HI results in EEG patterns similar to those observed in human neonates. Activation patterns revealed in this study help explain complex seizure behaviors and EEG patterns observed in neonatal HI injury. This pattern may be, in part, secondary to regional differences in development in the neonatal brain. ANN NEUROL 2019;86:927-938.
© 2019 American Neurological Association.
Conflict of interest statement
Potential Conflicts of Interest
Nothing to report.
Figures

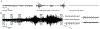




References
-
- Vasudevan C, Levene M. Epidemiology and aetiology of neonatal seizures. Semin Fetal Neonatal Med 2013;18:185–191. - PubMed
-
- Volpe J, Inder T, Darras B, et al. Neonatal seizures. In: Volpe’s neurology of the newborn. Amsterdam, the Netherlands: Elsevier, 2018: 275–321.
-
- Clancy RR. The contribution of EEG to the understanding of neonatal seizures. Epilepsia 2007;37(suppl 1):S52–S59. - PubMed
-
- Shellhaas RA, Clancy RR. Characterization of neonatal seizures by conventional EEG and single-channel EEG. Clin Neurophysiol 2007; 118:2156–2161. - PubMed
-
- Bye A, Flanagan D. Spatial and temporal characteristics of neonatal seizures. Epilepsia 1995;36:1009–1016. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

