Enlargement of a Modular System-Synthesis and Characterization of an s-Triazine-Based Carboxylic Acid Ester Bearing a Galactopyranosyl Moiety and an Enormous Boron Load
- PMID: 31509949
- PMCID: PMC6767515
- DOI: 10.3390/molecules24183288
Enlargement of a Modular System-Synthesis and Characterization of an s-Triazine-Based Carboxylic Acid Ester Bearing a Galactopyranosyl Moiety and an Enormous Boron Load
Abstract
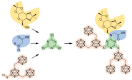
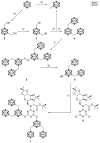
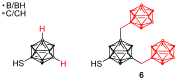
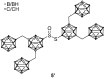
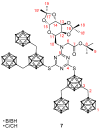
The amount of boron accumulated in tumor tissue plays an important role regarding the success of the boron neutron capture therapy (BNCT). In this article, we report a modular system, combining readily available starting materials, like glycine, 1,3,5-triazine and the well-known 9-mercapto-1,7-dicarba-closo-dodecaborane(12), as well as α-d-galactopyranose for increased hydrophilicity, with a novel boron-rich tris-meta-carboranyl thiol.
Keywords: boron neutron capture therapy; cancer therapy; carborane; modular system; s-triazine.
Conflict of interest statement
The authors declare the following competing financial interest(s): The work was partly supported by Bayer AG. This work was further supported by the federal state of Saxony, and the “European Regional Development Fund”.
Figures


Similar articles
-
Modular Synthetic Approach to Carboranyl‒Biomolecules Conjugates.Molecules. 2021 Apr 3;26(7):2057. doi: 10.3390/molecules26072057. Molecules. 2021. PMID: 33916755 Free PMC article.
-
Closomers of high boron content: synthesis, characterization, and potential application as unimolecular nanoparticle delivery vehicles for boron neutron capture therapy.Inorg Chem. 2006 Jan 9;45(1):278-85. doi: 10.1021/ic051214q. Inorg Chem. 2006. PMID: 16390066
-
Modular triazine-based carborane-containing carboxylic acids - synthesis and characterisation of potential boron neutron capture therapy agents made of readily accessible building blocks.Dalton Trans. 2019 Aug 7;48(29):10834-10844. doi: 10.1039/c9dt02130b. Epub 2019 Jun 27. Dalton Trans. 2019. PMID: 31246208
-
Boron Agent Delivery Platforms Based on Natural Products for Boron Neutron Capture Therapy.ChemMedChem. 2024 Nov 4;19(21):e202400323. doi: 10.1002/cmdc.202400323. Epub 2024 Oct 15. ChemMedChem. 2024. PMID: 38830821 Review.
-
Application of cycloaddition reactions to the syntheses of novel boron compounds.Molecules. 2010 Dec 21;15(12):9437-49. doi: 10.3390/molecules15129437. Molecules. 2010. PMID: 21178899 Free PMC article. Review.
Cited by
-
New Boron Delivery Agents.Cancer Biother Radiopharm. 2023 Apr;38(3):160-172. doi: 10.1089/cbr.2022.0060. Epub 2022 Nov 9. Cancer Biother Radiopharm. 2023. PMID: 36350709 Free PMC article. Review.
-
Nickel(II) and Palladium(II) Complexes with η5:κ1(N)-Coordinated Dicarbollide Ligands Containing Pendant Pyridine Group.Int J Mol Sci. 2023 Oct 11;24(20):15069. doi: 10.3390/ijms242015069. Int J Mol Sci. 2023. PMID: 37894752 Free PMC article.
-
Modular Synthetic Approach to Carboranyl‒Biomolecules Conjugates.Molecules. 2021 Apr 3;26(7):2057. doi: 10.3390/molecules26072057. Molecules. 2021. PMID: 33916755 Free PMC article.
References
-
- Locher G.L. Biological Effects and Therapeutic Possibilities of Neutrons. Am. J. Roentgenol. Radium Therapy. 1936;36:1–18.
-
- Hawthorne M.F. The Role of Chemistry in the Development of Boron Neutron Capture Therapy of Cancer. Angew. Chem. Int. Ed. 1993;32:950–984. doi: 10.1002/anie.199309501. - DOI
-
- Sivaev I.B., Bregadze V.V. Polyhedral Boranes for Medical Applications: Current Status and Perspectives. Eur. J. Inorg. Chem. 2009;2009:1433–1450. doi: 10.1002/ejic.200900003. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

