Monitoring of Low-Intensity Exposures via Luminescent Bioassays of Different Complexity: Cells, Enzyme Reactions, and Fluorescent Proteins
- PMID: 31509958
- PMCID: PMC6770735
- DOI: 10.3390/ijms20184451
Monitoring of Low-Intensity Exposures via Luminescent Bioassays of Different Complexity: Cells, Enzyme Reactions, and Fluorescent Proteins
Abstract
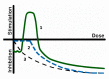
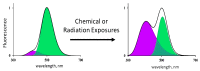
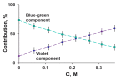
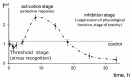
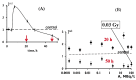
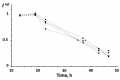
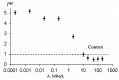
The current paper reviews the applications of luminescence bioassays for monitoring the results of low-intensity exposures which produce a stimulative effect. The impacts of radioactivity of different types (alpha, beta, and gamma) and bioactive compounds (humic substances and fullerenols) are under consideration. Bioassays based on luminous marine bacteria, their enzymes, and fluorescent coelenteramide-containing proteins were used to compare the results of the low-intensity exposures at the cellular, biochemical, and physicochemical levels, respectively. High rates of luminescence response can provide (1) a proper number of experimental results under comparable conditions and, therefore, proper statistical processing, with this being highly important for "noisy" low-intensity exposures; and (2) non-genetic, i.e., biochemical and physicochemical mechanisms of cellular response for short-term exposures. The results of cellular exposures were discussed in terms of the hormesis concept, which implies low-dose stimulation and high-dose inhibition of physiological functions. Dependencies of the luminescence response on the exposure time or intensity (radionuclide concentration/gamma radiation dose rate, concentration of the bioactive compounds) were analyzed and compared for bioassays of different organization levels.
Keywords: antioxidant activity; bacterial cells, enzymes; bioactive compounds; fluorescent protein; hormesis; low-intensity factors; luminescence bioassays; radiation.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the writing of the manuscript or in the decision to publish the results.
Figures




Similar articles
-
Marine Bacteria under Low-Intensity Radioactive Exposure: Model Experiments.Int J Mol Sci. 2022 Dec 27;24(1):410. doi: 10.3390/ijms24010410. Int J Mol Sci. 2022. PMID: 36613854 Free PMC article. Review.
-
Enzymatic Responses to Low-Intensity Radiation of Tritium.Int J Mol Sci. 2020 Nov 11;21(22):8464. doi: 10.3390/ijms21228464. Int J Mol Sci. 2020. PMID: 33187108 Free PMC article.
-
Humic Substances Mitigate the Impact of Tritium on Luminous Marine Bacteria. Involvement of Reactive Oxygen Species.Int J Mol Sci. 2020 Sep 16;21(18):6783. doi: 10.3390/ijms21186783. Int J Mol Sci. 2020. PMID: 32947870 Free PMC article.
-
Variability of fluorescence spectra of coelenteramide-containing proteins as a basis for toxicity monitoring.Talanta. 2017 Aug 1;170:425-431. doi: 10.1016/j.talanta.2017.04.043. Epub 2017 Apr 18. Talanta. 2017. PMID: 28501192
-
Effect of low-dose ionizing radiation on luminous marine bacteria: radiation hormesis and toxicity.J Environ Radioact. 2015 Apr;142:68-77. doi: 10.1016/j.jenvrad.2015.01.012. Epub 2015 Jan 30. J Environ Radioact. 2015. PMID: 25644753 Review.
Cited by
-
Antioxidant Supplements versus Health Benefits of Brief/Intermittent Exposure to Potentially Toxic Physical or Chemical Agents.Curr Issues Mol Biol. 2021 Jul 10;43(2):650-664. doi: 10.3390/cimb43020047. Curr Issues Mol Biol. 2021. PMID: 34287292 Free PMC article. Review.
-
Marine Bacteria under Low-Intensity Radioactive Exposure: Model Experiments.Int J Mol Sci. 2022 Dec 27;24(1):410. doi: 10.3390/ijms24010410. Int J Mol Sci. 2022. PMID: 36613854 Free PMC article. Review.
-
Enzymatic Responses to Low-Intensity Radiation of Tritium.Int J Mol Sci. 2020 Nov 11;21(22):8464. doi: 10.3390/ijms21228464. Int J Mol Sci. 2020. PMID: 33187108 Free PMC article.
-
Endohedral Gd-Containing Fullerenol: Toxicity, Antioxidant Activity, and Regulation of Reactive Oxygen Species in Cellular and Enzymatic Systems.Int J Mol Sci. 2022 May 5;23(9):5152. doi: 10.3390/ijms23095152. Int J Mol Sci. 2022. PMID: 35563539 Free PMC article.
-
Toxicity and Antioxidant Activity of Fullerenol C60,70 with Low Number of Oxygen Substituents.Int J Mol Sci. 2021 Jun 15;22(12):6382. doi: 10.3390/ijms22126382. Int J Mol Sci. 2021. PMID: 34203700 Free PMC article.
References
-
- Southam C.M., Ehrlich J. Effects of extracts of western red-cedar heartwood on certain wood-decaying fungi in culture. Phytopathology. 1943;33:517–524.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

