HNF1B Mutations Are Associated With a Gitelman-like Tubulopathy That Develops During Childhood
- PMID: 31517149
- PMCID: PMC6732753
- DOI: 10.1016/j.ekir.2019.05.019
HNF1B Mutations Are Associated With a Gitelman-like Tubulopathy That Develops During Childhood
Abstract
Background: Mutations in the transcription factor hepatocyte nuclear factor 1B (HNF1B) are the most common inherited cause of renal malformations, yet also associated with renal tubular dysfunction, most prominently magnesium wasting with hypomagnesemia. The presence of hypomagnesemia has been proposed to help select appropriate patients for genetic testing. Yet, in a large cohort, hypomagnesemia was discriminatory only in adult, but not in pediatric patients. We therefore investigated whether hypomagnesemia and other biochemical changes develop with age.
Methods: We performed a retrospective analysis of clinical, biochemical, and genetic results of pediatric patients with renal malformations tested for HNF1B mutations, separated into 4 age groups. Values were excluded if concurrent estimated glomerular filtration rate (eGFR) was <30 ml/min per 1.73 m2, or after transplantation.
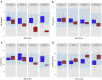
Results: A total of 199 patients underwent HNF1B genetic testing and mutations were identified in 52 (mut+). The eGFRs were comparable between mut+ and mut- in any age group. Although median plasma magnesium concentrations differed significantly between mut+ and mut- patients in all age groups, overt hypomagnesemia was not present until the second half of childhood in the mut+ group. There was also a significant difference in median potassium concentrations in late childhood with lower values in the mut+ cohort.
Conclusions: The abnormal tubular electrolyte handling associated with HNF1B mutations develops with age and is not restricted to magnesium, but consistent with a more generalized dysfunction of the distal convoluted tubule, reminiscent of Gitelman syndrome. The absence of these abnormalities in early childhood should not preclude HNF1B mutations from diagnostic considerations.
Keywords: HNF1B; alkalosis; children; hypokalemia; hypomagnesemia; renal tubular function.
Figures


References
-
- Coffinier C., Thepot D., Babinet C. Essential role for the homeoprotein vHNF1/HNF1beta in visceral endoderm differentiation. Development. 1999;126:4785–4794. - PubMed
-
- Eckardt K.U., Alper S.L., Antignac C. Autosomal dominant tubulointerstitial kidney disease: diagnosis, classification, and management—a KDIGO consensus report. Kidney Int. 2015;88:676–683. - PubMed
-
- Bockenhauer D., Jaureguiberry G. HNF1B-associated clinical phenotypes:the kidney and beyond. Pediatr Nephrol. 2016;31:707–714. - PubMed
-
- Clissold R.L., Hamilton A.J., Hattersley A.T. HNF1B-associated renal and extra-renal disease-an expanding clinical spectrum. Nat Rev Nephrol. 2015;11:102–112. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

