Effects of a single bout of exercise on mitochondria-mediated apoptotic signaling in rat cardiac and skeletal muscles
- PMID: 31523670
- PMCID: PMC6732537
- DOI: 10.12965/jer.1938380.190
Effects of a single bout of exercise on mitochondria-mediated apoptotic signaling in rat cardiac and skeletal muscles
Abstract
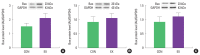
This study aimed to determine the effects of a single bout exercise on mitochondria-mediated apoptotic signaling in cardiac and skeletal muscles. Fischer 344 rats (4 months old) were randomly divided into the control or a single bout of exercise group (n=10 each). The rats performed a single bout of treadmill exercise for 60 min. Mitochondria-mediated apoptotic signaling (e.g., Bax, Bcl-2, mitochondrial permeability transition pore [mPTP] opening, cytochrome c, and cleaved caspase-3) was measured in cardiac (e.g., left ventricle) and skeletal (e.g., soleus and white gastrocnemius) muscles. A single bout of exercise significantly decreased mPTP opening sensitivity in all tissues. However, a single bout of exercise did not show any statistical differences in Bax, Bcl-2, cytochrome c, and cleaved caspase-3 in all tissues measured. A single bout of exercise did not show definite results on characteristics of mitochondria-mediated apoptotic signaling. Therefore, further research is necessary to provide a more mechanistic understanding of the apoptosis pathway.
Keywords: Apoptosis; Exercise; Heart; Mitochondria; Skeletal muscle.
Conflict of interest statement
CONFLICT OF INTEREST No potential conflict of interest relevant to this article was reported.
Figures





Similar articles
-
Effects of Acute Exercise on Mitochondrial Function, Dynamics, and Mitophagy in Rat Cardiac and Skeletal Muscles.Int Neurourol J. 2019 Feb;23(Suppl 1):S22-31. doi: 10.5213/inj.1938038.019. Epub 2019 Feb 28. Int Neurourol J. 2019. PMID: 30832464 Free PMC article.
-
Panax quinquefolium saponin attenuates cardiomyocyte apoptosis and opening of the mitochondrial permeability transition pore in a rat model of ischemia/reperfusion.Cell Physiol Biochem. 2014;34(4):1413-26. doi: 10.1159/000366347. Epub 2014 Oct 3. Cell Physiol Biochem. 2014. PMID: 25301366
-
Acute exercise protects against calcium-induced cardiac mitochondrial permeability transition pore opening in doxorubicin-treated rats.Clin Sci (Lond). 2011 Jan;120(1):37-49. doi: 10.1042/CS20100254. Clin Sci (Lond). 2011. PMID: 20666733
-
Effects of aging and exercise training on mitochondrial function and apoptosis in the rat heart.Pflugers Arch. 2020 Feb;472(2):179-193. doi: 10.1007/s00424-020-02357-6. Epub 2020 Feb 11. Pflugers Arch. 2020. PMID: 32048000
-
Mitochondria and cell death.Biochem Soc Trans. 2000 Feb;28(2):170-7. doi: 10.1042/bst0280170. Biochem Soc Trans. 2000. PMID: 10816121 Review.
Cited by
-
Treadmill Running Improves Spatial Learning Memory Through Inactivation of Nuclear Factor Kappa B/Mitogen-Activated Protein Kinase Signaling Pathway in Amyloid-β-Induced Alzheimer Disease Rats.Int Neurourol J. 2021 May;25(Suppl 1):S35-43. doi: 10.5213/inj.2142164.082. Epub 2021 May 31. Int Neurourol J. 2021. PMID: 34053209 Free PMC article.
-
SEZ6L2 knockdown impairs tumour growth by promoting caspase-dependent apoptosis in colorectal cancer.J Cell Mol Med. 2020 Apr;24(7):4223-4232. doi: 10.1111/jcmm.15082. Epub 2020 Feb 27. J Cell Mol Med. 2020. PMID: 32105413 Free PMC article.
-
Combined effects of natural products and exercise on apoptosis pathways in obesity-related skeletal muscle dysfunction.Apoptosis. 2025 Apr;30(3-4):537-552. doi: 10.1007/s10495-024-02069-7. Epub 2025 Jan 20. Apoptosis. 2025. PMID: 39833631 Review.
-
The effect of endurance exercise and rosehip extract supplementation on the expression of P53 and cytochrome C genes in male rat heart.J Cardiovasc Thorac Res. 2022;14(4):246-252. doi: 10.34172/jcvtr.2022.31599. Epub 2022 Dec 31. J Cardiovasc Thorac Res. 2022. PMID: 36699555 Free PMC article.
-
The cardioprotective effect of S. africana caerulea/Blue Sage in ischaemia and reperfusion induced oxidative stress.Front Pharmacol. 2023 Sep 25;14:1254561. doi: 10.3389/fphar.2023.1254561. eCollection 2023. Front Pharmacol. 2023. PMID: 37818190 Free PMC article.
References
-
- Boroujerdi SS, Rahimi R. The apoptotic response to resistance exercise with different intensities in athletes. Med Sport. 2011;64:31–44.
-
- Gleeson M, Bishop NC, Stensel DJ, Lindley MR, Mastana SS, Nimmo MA. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol. 2011;11:607–615. - PubMed
-
- Heo JW, No MH, Min DH, Kang JH, Kwak HB. Aging-induced sarcopenia and exercise. Asian J Kinesiol. 2017;19:43–59.
LinkOut - more resources
Full Text Sources
Research Materials

