Human motor fatigability as evoked by repetitive movements results from a gradual breakdown of surround inhibition
- PMID: 31524600
- PMCID: PMC6746551
- DOI: 10.7554/eLife.46750
Human motor fatigability as evoked by repetitive movements results from a gradual breakdown of surround inhibition
Abstract
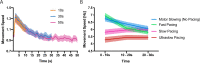
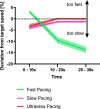
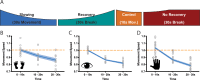
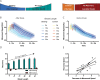
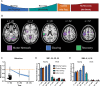
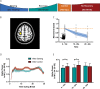
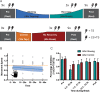
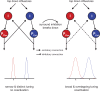
Motor fatigability emerges when demanding tasks are executed over an extended period of time. Here, we used repetitive low-force movements that cause a gradual reduction in movement speed (or 'motor slowing') to study the central component of fatigability in healthy adults. We show that motor slowing is associated with a gradual increase of net excitability in the motor network and, specifically, in primary motor cortex (M1), which results from overall disinhibition. Importantly, we link performance decrements to a breakdown of surround inhibition in M1, which is associated with high coactivation of antagonistic muscle groups. This is consistent with the model that a loss of inhibitory control might broaden the tuning of population vectors such that movement patterns become more variable, ill-timed and effortful. We propose that the release of inhibition in M1 is an important mechanism underpinning motor fatigability and, potentially, also pathological fatigue as frequently observed in patients with brain disorders.
Keywords: electrophysiology; fatigue; functional magnetic resonance imaging; human; motor slowing; neuroscience; repetitive movements; transcranial magnetic stimulation.
© 2019, Bächinger et al.
Conflict of interest statement
MB, RL, FT, SH, JB, NW No competing interests declared
Figures


References
-
- Arias P, Robles-García V, Corral-Bergantiños Y, Madrid A, Espinosa N, Valls-Solé J, Grieve KL, Oliviero A, Cudeiro J. Central fatigue induced by short-lasting finger tapping and isometric tasks: a study of silent periods evoked at spinal and supraspinal levels. Neuroscience. 2015;305:316–327. doi: 10.1016/j.neuroscience.2015.07.081. - DOI - PubMed
-
- Bächinger M, Zerbi V, Moisa M, Polania R, Liu Q, Mantini D, Ruff C, Wenderoth N. Concurrent tACS-fMRI reveals causal influence of power synchronized neural activity on resting state fMRI connectivity. The Journal of Neuroscience. 2017;37:4766–4777. doi: 10.1523/JNEUROSCI.1756-16.2017. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

