VX-770-mediated potentiation of numerous human CFTR disease mutants is influenced by phosphorylation level
- PMID: 31530897
- PMCID: PMC6749054
- DOI: 10.1038/s41598-019-49921-4
VX-770-mediated potentiation of numerous human CFTR disease mutants is influenced by phosphorylation level
Abstract
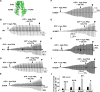
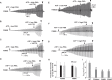
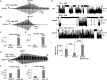
VX-770 (ivacaftor) is approved for clinical use in CF patients bearing multiple CFTR mutations. VX-770 potentiated wildtype CFTR and several disease mutants expressed in oocytes in a manner modulated by PKA-mediated phosphorylation. Potentiation of some other mutants, including G551D-CFTR, was less dependent upon the level of phosphorylation, likely related to the severe gating defects in these mutants exhibited in part by a shift in PKA sensitivity to activation, possibly due to an electrostatic interaction of D551 with K1250. Phosphorylation-dependent potentiation of wildtype CFTR and other variants also was observed in epithelial cells. Hence, the efficacy of potentiators may be obscured by a ceiling effect when drug screening is performed under strongly phosphorylating conditions. These results should be considered in campaigns for CFTR potentiator discovery, and may enable the expansion of VX-770 to CF patients bearing ultra-orphan CFTR mutations.
Conflict of interest statement
The authors declare no competing interests.
Figures







Similar articles
-
Mutation-specific dual potentiators maximize rescue of CFTR gating mutants.J Cyst Fibros. 2020 Mar;19(2):236-244. doi: 10.1016/j.jcf.2019.10.011. Epub 2019 Oct 31. J Cyst Fibros. 2020. PMID: 31678009 Free PMC article.
-
Cystic fibrosis transmembrane conductance regulator (CFTR) potentiators protect G551D but not ΔF508 CFTR from thermal instability.Biochemistry. 2014 Sep 9;53(35):5613-8. doi: 10.1021/bi501007v. Epub 2014 Aug 22. Biochemistry. 2014. PMID: 25148434 Free PMC article.
-
Synergistic Potentiation of Cystic Fibrosis Transmembrane Conductance Regulator Gating by Two Chemically Distinct Potentiators, Ivacaftor (VX-770) and 5-Nitro-2-(3-Phenylpropylamino) Benzoate.Mol Pharmacol. 2016 Sep;90(3):275-85. doi: 10.1124/mol.116.104570. Epub 2016 Jul 13. Mol Pharmacol. 2016. PMID: 27413118 Free PMC article.
-
Cystic fibrosis transmembrane conductance regulator-modifying medications: the future of cystic fibrosis treatment.Ann Pharmacother. 2012 Jul-Aug;46(7-8):1065-75. doi: 10.1345/aph.1R076. Epub 2012 Jun 26. Ann Pharmacother. 2012. PMID: 22739718 Review.
-
Lumacaftor and ivacaftor in the management of patients with cystic fibrosis: current evidence and future prospects.Ther Adv Respir Dis. 2015 Dec;9(6):313-26. doi: 10.1177/1753465815601934. Epub 2015 Sep 28. Ther Adv Respir Dis. 2015. PMID: 26416827 Review.
Cited by
-
A small molecule CFTR potentiator restores ATP-dependent channel gating to the cystic fibrosis mutant G551D-CFTR.Br J Pharmacol. 2022 Apr;179(7):1319-1337. doi: 10.1111/bph.15709. Epub 2022 Jan 21. Br J Pharmacol. 2022. PMID: 34644413 Free PMC article.
-
Antisense oligonucleotide-mediated correction of CFTR splicing improves chloride secretion in cystic fibrosis patient-derived bronchial epithelial cells.Nucleic Acids Res. 2020 Jul 27;48(13):7454-7467. doi: 10.1093/nar/gkaa490. Nucleic Acids Res. 2020. PMID: 32520327 Free PMC article.
-
Mutation-specific dual potentiators maximize rescue of CFTR gating mutants.J Cyst Fibros. 2020 Mar;19(2):236-244. doi: 10.1016/j.jcf.2019.10.011. Epub 2019 Oct 31. J Cyst Fibros. 2020. PMID: 31678009 Free PMC article.
-
The bidirectional relationship between CFTR and lipids.Commun Biol. 2020 Apr 20;3(1):179. doi: 10.1038/s42003-020-0909-1. Commun Biol. 2020. PMID: 32313074 Free PMC article. Review.
-
Two rare variants that affect the same amino acid in CFTR have distinct responses to ivacaftor.J Physiol. 2024 Jan;602(2):333-354. doi: 10.1113/JP285727. Epub 2024 Jan 7. J Physiol. 2024. PMID: 38186087 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

