Mechanosensing through immunoreceptors
- PMID: 31534240
- PMCID: PMC7592628
- DOI: 10.1038/s41590-019-0491-1
Mechanosensing through immunoreceptors
Erratum in
-
Author Correction: Mechanosensing through immunoreceptors.Nat Immunol. 2019 Dec;20(12):1700. doi: 10.1038/s41590-019-0545-4. Nat Immunol. 2019. PMID: 31686010
Abstract
The immune response is orchestrated by a variety of immune cells. The function of each cell is determined by the collective signals from various immunoreceptors, whose expression and activity depend on the developmental stages of the cell and its environmental context. Recent studies have highlighted the presence of mechanical force on several immunoreceptor-ligand pairs and the important role of force in regulating their interaction and function. In this Perspective, we use the T cell antigen receptor as an example with which to review the current understanding of the mechanosensing properties of immunoreceptors. We discuss the types of forces that immunoreceptors may encounter and the effects of force on ligand bonding, conformational change and the triggering of immunoreceptors, as well as the effects of force on the downstream signal transduction, cell-fate decisions and effector function of immune cells.
Conflict of interest statement
Competing interests
The authors declare no competing interests.
Figures
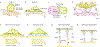

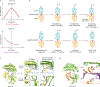
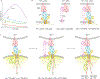
References
-
- Mempel TR, Henrickson SE & Von Andrian UH T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

