Mg Doped CuCrO2 as Efficient Hole Transport Layers for Organic and Perovskite Solar Cells
- PMID: 31540282
- PMCID: PMC6781018
- DOI: 10.3390/nano9091311
Mg Doped CuCrO2 as Efficient Hole Transport Layers for Organic and Perovskite Solar Cells
Abstract
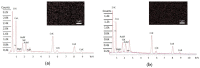
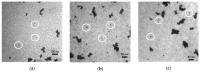
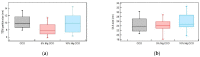
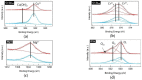
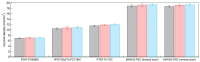
The electrical and optical properties of the hole transport layer (HTL) are critical for organic and halide perovskite solar cell (OSC and PSC, respectively) performance. In this work, we studied the effect of Mg doping on CuCrO2 (CCO) nanoparticles and their performance as HTLs in OSCs and PSCs. CCO and Mg doped CCO (Mg:CCO) nanoparticles were hydrothermally synthesized. The nanoparticles were characterized by various experimental techniques to study the effect of Mg doping on structural, chemical, morphological, optical, and electronic properties of CCO. We found that Mg doping increases work function and decreases particle size. We demonstrate CCO and Mg:CCO as efficient HTLs in a variety of OSCs, including the first demonstration of a non-fullerene acceptor bulk heterojunction, and CH3NH3PbI3 PSCs. A small improvement of average short-circuit current density with Mg doping was found in all systems.
Keywords: Mg doped CuCrO2; hole transport layer; organic solar cells; perovskite solar cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Advances in Hole Transport Materials for Layered Casting Solar Cells.Polymers (Basel). 2023 Nov 17;15(22):4443. doi: 10.3390/polym15224443. Polymers (Basel). 2023. PMID: 38006166 Free PMC article. Review.
-
Boosting the Conversion Efficiency Over 20% in MAPbI3 Perovskite Planar Solar Cells by Employing a Solution-Processed Aluminum-Doped Nickel Oxide Hole Collector.ACS Appl Mater Interfaces. 2020 May 20;12(20):22958-22970. doi: 10.1021/acsami.0c04618. Epub 2020 May 8. ACS Appl Mater Interfaces. 2020. PMID: 32326692
-
CuCrO2 Nanoparticles Incorporated into PTAA as a Hole Transport Layer for 85 °C and Light Stabilities in Perovskite Solar Cells.Nanomaterials (Basel). 2020 Aug 26;10(9):1669. doi: 10.3390/nano10091669. Nanomaterials (Basel). 2020. PMID: 32858913 Free PMC article.
-
High-Efficiency and Stable Inverted Planar Perovskite Solar Cells with Pulsed Laser Deposited Cu-Doped NiOx Hole-Transport Layers.ACS Appl Mater Interfaces. 2020 Nov 11;12(45):50684-50691. doi: 10.1021/acsami.0c15923. Epub 2020 Oct 29. ACS Appl Mater Interfaces. 2020. PMID: 33121249
-
Recent Advances in Nanostructured Inorganic Hole-Transporting Materials for Perovskite Solar Cells.Nanomaterials (Basel). 2022 Jul 28;12(15):2592. doi: 10.3390/nano12152592. Nanomaterials (Basel). 2022. PMID: 35957022 Free PMC article. Review.
Cited by
-
Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells.Nanomaterials (Basel). 2021 Aug 19;11(8):2109. doi: 10.3390/nano11082109. Nanomaterials (Basel). 2021. PMID: 34443938 Free PMC article.
-
Improving the Performance of Polymer Solar Cells with Benzo[ghi]perylenetriimide-Based Small-Molecules as Interfacial Layers.Polymers (Basel). 2022 Oct 21;14(20):4466. doi: 10.3390/polym14204466. Polymers (Basel). 2022. PMID: 36298044 Free PMC article.
-
Solution Synthesis, Processing, and Applications of Semiconducting Nanomaterials.Nanomaterials (Basel). 2019 Oct 11;9(10):1442. doi: 10.3390/nano9101442. Nanomaterials (Basel). 2019. PMID: 31614524 Free PMC article.
-
Advances in Hole Transport Materials for Layered Casting Solar Cells.Polymers (Basel). 2023 Nov 17;15(22):4443. doi: 10.3390/polym15224443. Polymers (Basel). 2023. PMID: 38006166 Free PMC article. Review.
-
Opportunities and challenges of hole transport materials for high-performance inverted hybrid-perovskite solar cells.Exploration (Beijing). 2023 Feb 24;3(3):20220027. doi: 10.1002/EXP.20220027. eCollection 2023 Jun. Exploration (Beijing). 2023. PMID: 37933381 Free PMC article.
References
-
- Yip H.-L., Jen A.K.-Y. Recent advances in solution-processed interfacial materials for efficient and stable polymer solar cells. Energy Environ. Sci. 2012;5:5994–6011. doi: 10.1039/c2ee02806a. - DOI
-
- Yan C., Barlow S., Wang Z., Yan H., Jen A.K.-Y., Marder S.R., Zhan X. Non-fullerene acceptors for organic solar cells. Nat. Rev. Mater. 2018;3:18003. doi: 10.1038/natrevmats.2018.3. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

