Dysregulation of Hyaluronan Homeostasis During White Matter Injury
- PMID: 31542857
- PMCID: PMC7060835
- DOI: 10.1007/s11064-019-02879-1
Dysregulation of Hyaluronan Homeostasis During White Matter Injury
Abstract
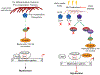
Although the extra cellular matrix (ECM) comprises a major proportion of the CNS parenchyma, new roles for the ECM in regeneration and repair responses to CNS injury have only recently been appreciated. The ECM undergoes extensive remodeling following injury to the developing or mature CNS in disorders that -include perinatal hypoxic-ischemic cerebral injury, multiple sclerosis and age-related vascular dementia. Here we focus on recently described mechanisms involving hyaluronan (HA), which negatively impact myelin repair after cerebral white matter injury. Injury induced depolymerization of hyaluronan (HA)-a component of the neural ECM-can inhibit myelin repair through the actions of specific sizes of HA fragments. These bioactive fragments selectively block the maturation of late oligodendrocyte progenitors via an immune tolerance-like pathway that suppresses pro-myelination signaling. We highlight emerging new pathophysiological roles of the neural ECM, particularly of those played by HA fragments (HAf) after injury and discuss strategies to promoter repair and regeneration of chronic myelination failure.
Keywords: Extracellular matrix; Hyaluronan; Neuroinflammation; White matter injury.
Conflict of interest statement
Conflicts of Interest
The authors declare no conflicts of interests.
Figures
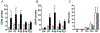
References
-
- Bignami A, Hosley M, Dahl D, Hyaluronic acid and hyaluronic acid-binding proteins in brain extracellular matrix. Anat Embryol (Berl) 188, 419–433 (1993). - PubMed
-
- Cragg B, Brain extracellular space fixed for electron microscopy. Neurosci Lett 15, 301–306 (1979). - PubMed
-
- Lau LW, Cua R, Keough MB, Haylock-Jacobs S, Yong VW, Pathophysiology of the brain extracellular matrix: a new target for remyelination. Nat Rev Neurosci 14, 722–729 (2013). - PubMed
-
- Bandtlow CE, Zimmermann DR, Proteoglycans in the developing brain: new conceptual insights for old proteins. Physiol Rev 80, 1267–1290 (2000). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS054044/NS/NINDS NIH HHS/United States
- P51 OD011092/OD/NIH HHS/United States
- R01 NS105984/NS/NINDS NIH HHS/United States
- NS054044/NS/NINDS NIH HHS/United States
- R01 AG031892/AG/NIA NIH HHS/United States
- RF1 AG065406/AG/NIA NIH HHS/United States
- 11GRNT7510072/American Heart Association
- RG4843A5/1/National Multiple Sclerosis Society
- R01 NS116674/NS/NINDS NIH HHS/United States
- MS160144/Congressionally Directed Medical Research Programs
- R01 NS045737/NS/NINDS NIH HHS/United States
- R37 NS045737/NS/NINDS NIH HHS/United States
- NS045737/NS/NINDS NIH HHS/United States
- AG-31892/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources

