Zinc Maintains Embryonic Stem Cell Pluripotency and Multilineage Differentiation Potential via AKT Activation
- PMID: 31544103
- PMCID: PMC6728745
- DOI: 10.3389/fcell.2019.00180
Zinc Maintains Embryonic Stem Cell Pluripotency and Multilineage Differentiation Potential via AKT Activation
Abstract
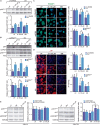
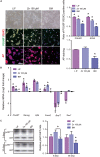
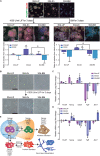
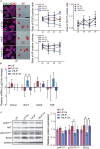
Embryonic stem cells (ESCs) possess remarkable abilities, as they can differentiate into all cell types (pluripotency) and be self-renewing, giving rise to two identical cells. These characteristics make ESCs a powerful research tool in fundamental embryogenesis as well as candidates for use in regenerative medicine. Significant efforts have been devoted to developing protocols to control ESC fate, including soluble and complex cocktails of growth factors and small molecules seeking to activate/inhibit key signaling pathways for the maintenance of pluripotency states or activate differentiation. Here we describe a novel method for the effective maintenance of mouse ESCs, avoiding the supplementation of complex inhibitory cocktails or cytokines, e.g., LIF. We show that the addition of zinc to ESC cultures leads to a stable pluripotent state that shares biochemical, transcriptional and karyotypic features with the classical LIF treatment. We demonstrate for the first time that ESCs maintained in long-term cultures with added zinc, are capable of sustaining a stable ESCs pluripotent phenotype, as well as differentiating efficiently upon external stimulation. We show that zinc promotes long-term ESC self-renewal (>30 days) via activation of ZIP7 and AKT signaling pathways. Furthermore, the combination of zinc with LIF results in a synergistic effect that enhances LIF effects, increases AKT and STAT3 activity, promotes the expression of pluripotency regulators and avoids the expression of differentiation markers.
Keywords: AKT; ZIP7; embryonic stem cells (ESC); stemness maintenance; zinc.
Figures



Similar articles
-
Pramel7 mediates LIF/STAT3-dependent self-renewal in embryonic stem cells.Stem Cells. 2011 Mar;29(3):474-85. doi: 10.1002/stem.588. Stem Cells. 2011. PMID: 21425410
-
Promotion of feeder-independent self-renewal of embryonic stem cells by retinol (vitamin A).Stem Cells. 2008 Jul;26(7):1858-64. doi: 10.1634/stemcells.2008-0050. Epub 2008 Apr 24. Stem Cells. 2008. PMID: 18436859
-
Retinoic acid maintains self-renewal of murine embryonic stem cells via a feedback mechanism.Differentiation. 2008 Nov;76(9):931-45. doi: 10.1111/j.1432-0436.2008.00272.x. Epub 2008 Jul 1. Differentiation. 2008. PMID: 18637026
-
Regulation of Embryonic Stem Cell Self-Renewal.Life (Basel). 2022 Jul 29;12(8):1151. doi: 10.3390/life12081151. Life (Basel). 2022. PMID: 36013330 Free PMC article. Review.
-
LIF signal in mouse embryonic stem cells.JAKSTAT. 2015 Sep 11;4(2):e1086520. doi: 10.1080/21623996.2015.1086520. eCollection 2015. JAKSTAT. 2015. PMID: 27127728 Free PMC article. Review.
Cited by
-
Impact of Zinc Transport Mechanisms on Embryonic and Brain Development.Nutrients. 2022 Jun 17;14(12):2526. doi: 10.3390/nu14122526. Nutrients. 2022. PMID: 35745255 Free PMC article. Review.
-
Zinc enhances the cell adhesion, migration, and self-renewal potential of human umbilical cord derived mesenchymal stem cells.World J Stem Cells. 2023 Jul 26;15(7):751-767. doi: 10.4252/wjsc.v15.i7.751. World J Stem Cells. 2023. PMID: 37545753 Free PMC article.
-
Metal transporter Slc30a1 controls pharyngeal neural crest differentiation via the zinc-Snai2-Jag1 cascade.MedComm (2020). 2021 Sep 27;2(4):778-797. doi: 10.1002/mco2.91. eCollection 2021 Dec. MedComm (2020). 2021. PMID: 34977877 Free PMC article.
-
Strontium-Modified Scaffolds Based on Mesoporous Bioactive Glasses/Polyvinyl Alcohol Composites for Bone Regeneration.Materials (Basel). 2020 Dec 3;13(23):5526. doi: 10.3390/ma13235526. Materials (Basel). 2020. PMID: 33287381 Free PMC article.
References
-
- Armstrong L., Hughes O., Yung S., Hyslop L., Stewart R., Wappler I., et al. (2006). The role of PI3K/AKT, MAPK/ERK and NFκβ signalling in the maintenance of human embryonic stem cell pluripotency and viability highlighted by transcriptional profiling and functional analysis. Hum. Mol. Genet. 15 1894–1913. 10.1093/hmg/ddl112 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

