Incorporating new approach methodologies in toxicity testing and exposure assessment for tiered risk assessment using the RISK21 approach: Case studies on food contact chemicals
- PMID: 31545997
- PMCID: PMC7429715
- DOI: 10.1016/j.fct.2019.110819
Incorporating new approach methodologies in toxicity testing and exposure assessment for tiered risk assessment using the RISK21 approach: Case studies on food contact chemicals
Abstract
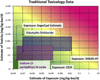
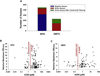
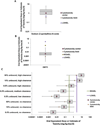
Programs including the ToxCast project have generated large amounts of in vitro high‒throughput screening (HTS) data, and best approaches for the interpretation and use of HTS data, including for chemical safety assessment, remain to be evaluated. To fill this gap, we conducted case studies of two indirect food additive chemicals where ToxCast data were compared with in vivo toxicity data using the RISK21 approach. Two food contact substances, sodium (2-pyridylthio)-N-oxide and dibutyltin dichloride, were selected, and available exposure data, toxicity data, and model predictions were compiled and assessed. Oral equivalent doses for the ToxCast bioactivity data were determined by in-vitro in-vivo extrapolation (IVIVE). For sodium (2-pyridylthio)-N-oxide, bioactive concentrations in ToxCast assays corresponded to low- and no-observed adverse effect levels in animal studies. For dibutyltin dichloride, the ToxCast bioactive concentrations were below the dose range that demonstrated toxicity in animals; however, this was confounded by the lack of toxicokinetic data, necessitating the use of conservative toxicokinetic parameter estimates for IVIVE calculations. This study highlights the potential utility of the RISK21 approach for interpretation of the ToxCast HTS data, as well as the challenges involved in integrating in vitro HTS data into safety assessments.
Keywords: Dibutyltin dichloride; Food additive; High-throughput screening; In-vitro in-vivo extrapolation; RISK21; Sodium (2-pyridylthio)-N-Oxide (pyrithione sodium); ToxCast.
Copyright © 2019 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures

Similar articles
-
Incorporating High-Throughput Exposure Predictions With Dosimetry-Adjusted In Vitro Bioactivity to Inform Chemical Toxicity Testing.Toxicol Sci. 2015 Nov;148(1):121-36. doi: 10.1093/toxsci/kfv171. Epub 2015 Aug 6. Toxicol Sci. 2015. PMID: 26251325 Free PMC article.
-
Evaluation of food-relevant chemicals in the ToxCast high-throughput screening program.Food Chem Toxicol. 2016 Jun;92:188-96. doi: 10.1016/j.fct.2016.04.012. Epub 2016 Apr 19. Food Chem Toxicol. 2016. PMID: 27103583
-
Potential of ToxCast Data in the Safety Assessment of Food Chemicals.Toxicol Sci. 2020 Apr 1;174(2):326-340. doi: 10.1093/toxsci/kfaa008. Toxicol Sci. 2020. PMID: 32040188 Free PMC article.
-
Illustrative case using the RISK21 roadmap and matrix: prioritization for evaluation of chemicals found in drinking water.Crit Rev Toxicol. 2016;46(1):43-53. doi: 10.3109/10408444.2015.1082973. Epub 2015 Oct 9. Crit Rev Toxicol. 2016. PMID: 26451723 Free PMC article. Review.
-
In vitro and modelling approaches to risk assessment from the U.S. Environmental Protection Agency ToxCast programme.Basic Clin Pharmacol Toxicol. 2014 Jul;115(1):69-76. doi: 10.1111/bcpt.12239. Epub 2014 Apr 22. Basic Clin Pharmacol Toxicol. 2014. PMID: 24684691 Review.
Cited by
-
Sourcing data on chemical properties and hazard data from the US-EPA CompTox Chemicals Dashboard: A practical guide for human risk assessment.Environ Int. 2021 Sep;154:106566. doi: 10.1016/j.envint.2021.106566. Epub 2021 Apr 29. Environ Int. 2021. PMID: 33934018 Free PMC article. Review.
-
Prediction of the packaging chemical migration into food and water by cutting-edge machine learning techniques.Sci Rep. 2025 Mar 6;15(1):7806. doi: 10.1038/s41598-025-92459-x. Sci Rep. 2025. PMID: 40050416 Free PMC article.
-
Human Chemical Exposure from Background Emissions in the United States and the Implication for Quantifying Risks from Marginal Emission Increase.Toxics. 2021 Nov 15;9(11):308. doi: 10.3390/toxics9110308. Toxics. 2021. PMID: 34822699 Free PMC article.
-
Evaluating consumer exposure to disinfecting chemicals against coronavirus disease 2019 (COVID-19) and associated health risks.Environ Int. 2020 Dec;145:106108. doi: 10.1016/j.envint.2020.106108. Epub 2020 Sep 3. Environ Int. 2020. PMID: 32927283 Free PMC article.
-
Prioritization of chemicals in food for risk assessment by integrating exposure estimates and new approach methodologies: A next generation risk assessment case study.Front Toxicol. 2022 Sep 19;4:933197. doi: 10.3389/ftox.2022.933197. eCollection 2022. Front Toxicol. 2022. PMID: 36199824 Free PMC article.
References
-
- Ali AA, Upreti RK, Kidwai AM, 1990. Assessment of di-and tri-butyltin interaction with skeletal muscle membranes agents. Bull. Environ. Contam. Toxicol 44, 29–38. - PubMed
-
- Barter Z, Bayliss M, Beaune P, Boobis A, Carlile D, Edwards R, Brian Houston J, Lake B, Lipscomb J, Pelkonen O, Tucke G, Rostami-Hodjegan A, 2007. Scaling factors for the extrapolation of in vivo metabolic Drug clearance from in vitro data: reaching a consensus on values of human micro-somal protein and hepatocellularity per gram of liver. Curr. Drug Metabol 8, 33–45. 10.2174/138920007779315053. - DOI - PubMed
-
- ChemicalBook, 2016. Chembook. [WWW Document]. URL. https://www.chemicalbook.com/.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

