Magnetic Nanoclusters Coated with Albumin, Casein, and Gelatin: Size Tuning, Relaxivity, Stability, Protein Corona, and Application in Nuclear Magnetic Resonance Immunoassay
- PMID: 31546937
- PMCID: PMC6781099
- DOI: 10.3390/nano9091345
Magnetic Nanoclusters Coated with Albumin, Casein, and Gelatin: Size Tuning, Relaxivity, Stability, Protein Corona, and Application in Nuclear Magnetic Resonance Immunoassay
Abstract
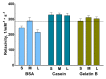
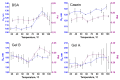
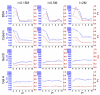
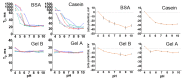
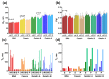
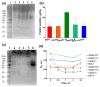
The surface functionalization of magnetic nanoparticles improves their physicochemical properties and applicability in biomedicine. Natural polymers, including proteins, are prospective coatings capable of increasing the stability, biocompatibility, and transverse relaxivity (r2) of magnetic nanoparticles. In this work, we functionalized the nanoclusters of carbon-coated iron nanoparticles with four proteins: bovine serum albumin, casein, and gelatins A and B, and we conducted a comprehensive comparative study of their properties essential to applications in biosensing. First, we examined the influence of environmental parameters on the size of prepared nanoclusters and synthesized protein-coated nanoclusters with a tunable size. Second, we showed that protein coating does not significantly influence the r2 relaxivity of clustered nanoparticles; however, the uniform distribution of individual nanoparticles inside the protein coating facilitates increased relaxivity. Third, we demonstrated the applicability of the obtained nanoclusters in biosensing by the development of a nuclear-magnetic-resonance-based immunoassay for the quantification of antibodies against tetanus toxoid. Fourth, the protein coronas of nanoclusters were studied using SDS-PAGE and Bradford protein assay. Finally, we compared the colloidal stability at various pH values and ionic strengths and in relevant complex media (i.e., blood serum, plasma, milk, juice, beer, and red wine), as well as the heat stability, resistance to proteolytic digestion, and shelf-life of protein-coated nanoclusters.
Keywords: antibody; assay; colloidal stability; nanoparticles; protein; protein G; streptavidin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Huang Z., Hu S., Xiong Y., Wei H., Xu H., Duan H., Lai W. Application and development of superparamagnetic nanoparticles in sample pretreatment and immunochromatographic assay. Trends Anal. Chem. 2019;114:151–170. doi: 10.1016/j.trac.2019.03.004. - DOI
-
- Rocha-Santos T.A.P. Sensors and biosensors based on magnetic nanoparticles. Trends Anal. Chem. 2014;62:28–36. doi: 10.1016/j.trac.2014.06.016. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

