Ca2+ Signaling in Cardiac Fibroblasts and Fibrosis-Associated Heart Diseases
- PMID: 31547577
- PMCID: PMC6956282
- DOI: 10.3390/jcdd6040034
Ca2+ Signaling in Cardiac Fibroblasts and Fibrosis-Associated Heart Diseases
Abstract
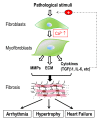
Cardiac fibrosis is the excessive deposition of extracellular matrix proteins by cardiac fibroblasts and myofibroblasts, and is a hallmark feature of most heart diseases, including arrhythmia, hypertrophy, and heart failure. This maladaptive process occurs in response to a variety of stimuli, including myocardial injury, inflammation, and mechanical overload. There are multiple signaling pathways and various cell types that influence the fibrogenesis cascade. Fibroblasts and myofibroblasts are central effectors. Although it is clear that Ca2+ signaling plays a vital role in this pathological process, what contributes to Ca2+ signaling in fibroblasts and myofibroblasts is still not wholly understood, chiefly because of the large and diverse number of receptors, transporters, and ion channels that influence intracellular Ca2+ signaling. Intracellular Ca2+ signals are generated by Ca2+ release from intracellular Ca2+ stores and by Ca2+ entry through a multitude of Ca2+-permeable ion channels in the plasma membrane. Over the past decade, the transient receptor potential (TRP) channels have emerged as one of the most important families of ion channels mediating Ca2+ signaling in cardiac fibroblasts. TRP channels are a superfamily of non-voltage-gated, Ca2+-permeable non-selective cation channels. Their ability to respond to various stimulating cues makes TRP channels effective sensors of the many different pathophysiological events that stimulate cardiac fibrogenesis. This review focuses on the mechanisms of Ca2+ signaling in fibroblast differentiation and fibrosis-associated heart diseases and will highlight recent advances in the understanding of the roles that TRP and other Ca2+-permeable channels play in cardiac fibrosis.
Keywords: Ca2+ signaling pathways; TRP channels; cardiac fibroblasts; cardiac fibrosis; ion channels.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Weber K. Cardiac interstitium. In: Poole-Wilson P., Colucci W., Massie B., Chatterjee K., Coats A., editors. Heart Failure. Churchill Livingstone; New York, NY, USA: 1997. pp. 13–31.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

