Simultaneous EEG-NIRS Measurement of the Inferior Parietal Lobule During a Reaching Task With Delayed Visual Feedback
- PMID: 31555114
- PMCID: PMC6742712
- DOI: 10.3389/fnhum.2019.00301
Simultaneous EEG-NIRS Measurement of the Inferior Parietal Lobule During a Reaching Task With Delayed Visual Feedback
Abstract
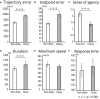
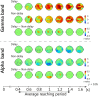
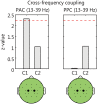
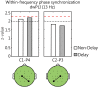
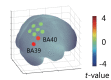
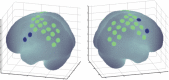
We investigated whether the inferior parietal lobule (IPL) responds in real-time to multisensory inconsistency during movement. The IPL is thought to be involved in both the detection of inconsistencies in multisensory information obtained during movement and that obtained during self-other discrimination. However, because of the limited temporal resolution of conventional neuroimaging techniques, it is difficult to distinguish IPL activity during movement from that during self-other discrimination. We simultaneously conducted electroencephalography (EEG) and near-infrared spectroscopy (NIRS) with the goal of examining IPL activity with a high spatiotemporal resolution during single reaching movements. Under a visual feedback-delay condition, gamma event-related synchronization (γ-ERS), i.e., an increase in gamma (31-47 Hz) EEG power occurred during reaching movements. This γ-ERS is considered to reflect processing of information about prediction errors. To integrate this temporal information with spatial information from the NIRS signals, we developed a new analysis technique that enabled estimation of the regions that show a hemodynamic response characterized by EEG fluctuation present in the visual feedback-delay condition. As a result, IPL activity was explained by γ-ERS specific to visual feedback delay during movements. Thus, we succeeded in demonstrating real-time activation of the IPL in response to multisensory inconsistency. However, we did not find any correlation between either IPL activity or γ-ERS with the sense of agency. Therefore, our results suggest that while the IPL is influenced by prediction error signals, it does not engage in direct processing underlying the conscious experience of making a movement, which is the foundation of self-other discrimination.
Keywords: electroencephalography; event-related (de)synchronization; inferior parietal lobule; near-infrared spectroscopy; psychophysiological interaction; simultaneous measurement; visual feedback delay.
Figures



Similar articles
-
Intracerebral ERD/ERS in voluntary movement and in cognitive visuomotor task.Prog Brain Res. 2006;159:311-30. doi: 10.1016/S0079-6123(06)59021-1. Prog Brain Res. 2006. PMID: 17071240
-
Functional mapping of human sensorimotor cortex with electrocorticographic spectral analysis. II. Event-related synchronization in the gamma band.Brain. 1998 Dec;121 ( Pt 12):2301-15. doi: 10.1093/brain/121.12.2301. Brain. 1998. PMID: 9874481
-
Distortion of Visuo-Motor Temporal Integration in Apraxia: Evidence From Delayed Visual Feedback Detection Tasks and Voxel-Based Lesion-Symptom Mapping.Front Neurol. 2018 Aug 27;9:709. doi: 10.3389/fneur.2018.00709. eCollection 2018. Front Neurol. 2018. PMID: 30210434 Free PMC article.
-
Functional anatomy of the macaque temporo-parieto-frontal connectivity.Cortex. 2017 Dec;97:306-326. doi: 10.1016/j.cortex.2016.12.007. Epub 2016 Dec 18. Cortex. 2017. PMID: 28041615 Review.
-
Schizophrenia and the inferior parietal lobule.Schizophr Res. 2007 Dec;97(1-3):215-25. doi: 10.1016/j.schres.2007.08.023. Epub 2007 Sep 12. Schizophr Res. 2007. PMID: 17851044 Review.
Cited by
-
Integration of the Cortical Haemodynamic Response Measured by Functional Near-Infrared Spectroscopy and Amino Acid Analysis to Aid in the Diagnosis of Major Depressive Disorder.Diagnostics (Basel). 2021 Oct 25;11(11):1978. doi: 10.3390/diagnostics11111978. Diagnostics (Basel). 2021. PMID: 34829325 Free PMC article.
-
Hybrid EEG-fNIRS BCI Fusion Using Multi-Resolution Singular Value Decomposition (MSVD).Front Hum Neurosci. 2020 Dec 8;14:599802. doi: 10.3389/fnhum.2020.599802. eCollection 2020. Front Hum Neurosci. 2020. PMID: 33363459 Free PMC article.
-
Enhanced spatiotemporal resolution imaging of neuronal activity using joint electroencephalography and diffuse optical tomography.Neurophotonics. 2021 Jan;8(1):015002. doi: 10.1117/1.NPh.8.1.015002. Epub 2021 Jan 1. Neurophotonics. 2021. PMID: 33437847 Free PMC article.
-
Analysis of Human Gait Using Hybrid EEG-fNIRS-Based BCI System: A Review.Front Hum Neurosci. 2021 Jan 25;14:613254. doi: 10.3389/fnhum.2020.613254. eCollection 2020. Front Hum Neurosci. 2021. PMID: 33568979 Free PMC article. Review.
-
Assessing social anhedonia in a transdiagnostic sample: Insights from a computational psychiatry lens.J Mood Anxiety Disord. 2024 Sep 17;8:100088. doi: 10.1016/j.xjmad.2024.100088. eCollection 2024 Dec. J Mood Anxiety Disord. 2024. PMID: 40655908 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials

