BET Proteins Are Required for Transcriptional Activation of the Senescent Islet Cell Secretome in Type 1 Diabetes
- PMID: 31561444
- PMCID: PMC6801956
- DOI: 10.3390/ijms20194776
BET Proteins Are Required for Transcriptional Activation of the Senescent Islet Cell Secretome in Type 1 Diabetes
Abstract
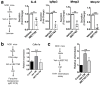
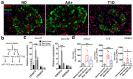
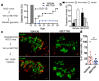
Type 1 diabetes (T1D) results from the progressive loss of pancreatic beta cells as a result of autoimmune destruction. We recently reported that during the natural history of T1D in humans and the female nonobese diabetic (NOD) mouse model, beta cells acquire a senescence-associated secretory phenotype (SASP) that is a major driver of disease onset and progression, but the mechanisms that activate SASP in beta cells were not explored. Here, we show that the SASP in islet cells is transcriptionally controlled by Bromodomain ExtraTerminal (BET) proteins, including Bromodomain containing protein 4 (BRD4). A chromatin analysis of key beta cell SASP genes in NOD islets revealed binding of BRD4 at active regulatory regions. BET protein inhibition in NOD islets diminished not only the transcriptional activation and secretion of SASP factors, but also the non-cell autonomous activity. BET protein inhibition also decreased the extent of SASP induction in human islets exposed to DNA damage. The BET protein inhibitor iBET-762 prevented diabetes in NOD mice and also attenuated SASP in islet cells in vivo. Taken together, our findings support a crucial role for BET proteins in the activation of the SASP transcriptional program in islet cells. These studies suggest avenues for preventing T1D by transcriptional inhibition of SASP.
Keywords: BET proteins; beta cells; senescence and SASP; type 1 diabetes.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Exploring senescence as a modifier of β cell extracellular vesicles in type 1 diabetes.Front Endocrinol (Lausanne). 2024 Aug 22;15:1422279. doi: 10.3389/fendo.2024.1422279. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39239092 Free PMC article. Review.
-
Exploring Transcriptional Regulation of Beta Cell SASP by Brd4-Associated Proteins and Cell Cycle Control Protein p21.Epigenomes. 2024 Mar 6;8(1):10. doi: 10.3390/epigenomes8010010. Epigenomes. 2024. PMID: 38534794 Free PMC article.
-
Secreted GDF15 maintains transcriptional responses during DNA damage-mediated senescence in human beta cells.Am J Physiol Endocrinol Metab. 2024 Oct 1;327(4):E552-E562. doi: 10.1152/ajpendo.00257.2024. Epub 2024 Aug 28. Am J Physiol Endocrinol Metab. 2024. PMID: 39196800
-
Pancreatic Alpha-Cells Contribute Together With Beta-Cells to CXCL10 Expression in Type 1 Diabetes.Front Endocrinol (Lausanne). 2020 Sep 15;11:630. doi: 10.3389/fendo.2020.00630. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 33042009 Free PMC article.
-
Type I diabetes mellitus: a predictable autoimmune disease with interindividual variation in the rate of beta cell destruction.Clin Immunol Immunopathol. 1989 Jan;50(1 Pt 2):S85-95. doi: 10.1016/0090-1229(89)90115-3. Clin Immunol Immunopathol. 1989. PMID: 2642771 Review.
Cited by
-
Exploring senescence as a modifier of β cell extracellular vesicles in type 1 diabetes.Front Endocrinol (Lausanne). 2024 Aug 22;15:1422279. doi: 10.3389/fendo.2024.1422279. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39239092 Free PMC article. Review.
-
Subcellular structure, heterogeneity, and plasticity of senescent cells.Aging Cell. 2024 Apr;23(4):e14154. doi: 10.1111/acel.14154. Epub 2024 Mar 30. Aging Cell. 2024. PMID: 38553952 Free PMC article. Review.
-
Exploring Transcriptional Regulation of Beta Cell SASP by Brd4-Associated Proteins and Cell Cycle Control Protein p21.Epigenomes. 2024 Mar 6;8(1):10. doi: 10.3390/epigenomes8010010. Epigenomes. 2024. PMID: 38534794 Free PMC article.
-
Polycomb Repressive Complexes: Shaping Pancreatic Beta-Cell Destiny in Development and Metabolic Disease.Front Cell Dev Biol. 2022 May 4;10:868592. doi: 10.3389/fcell.2022.868592. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35602600 Free PMC article. Review.
-
BRD4/MAP2K7/PGF Signaling Axis Promotes Senescence and Extracellular Matrix Metabolism of Nucleus Pulposus Cells in Intervertebral Disk Degeneration.Aging Cell. 2025 Jun;24(6):e70034. doi: 10.1111/acel.70034. Epub 2025 Mar 25. Aging Cell. 2025. PMID: 40130513 Free PMC article.
References
-
- Coppé J.-P., Patil C.K., Rodier F., Sun Y., Muñoz D.P., Goldstein J., Nelson P.S., Desprez P.-Y., Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi: 10.1371/journal.pbio.0060301. - DOI - PMC - PubMed

