The Effect of Abemaciclib Plus Fulvestrant on Overall Survival in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer That Progressed on Endocrine Therapy-MONARCH 2: A Randomized Clinical Trial
- PMID: 31563959
- PMCID: PMC6777264
- DOI: 10.1001/jamaoncol.2019.4782
The Effect of Abemaciclib Plus Fulvestrant on Overall Survival in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer That Progressed on Endocrine Therapy-MONARCH 2: A Randomized Clinical Trial
Abstract
Importance: Statistically significant overall survival (OS) benefits of CDK4 and CDK6 inhibitors in combination with fulvestrant for hormone receptor (HR)-positive, ERBB2 (formerly HER2)-negative advanced breast cancer (ABC) in patients regardless of menopausal status after prior endocrine therapy (ET) has not yet been demonstrated.
Objective: To compare the effect of abemaciclib plus fulvestrant vs placebo plus fulvestrant on OS at the prespecified interim of MONARCH 2 (338 events) in patients with HR-positive, ERBB2-negative advanced breast cancer that progressed during prior ET.
Design, setting, and participants: MONARCH 2 was a global, randomized, placebo-controlled, double-blind phase 3 trial of abemaciclib plus fulvestrant vs placebo plus fulvestrant for treatment of premenopausal or perimenopausal women (with ovarian suppression) and postmenopausal women with HR-positive, ERBB2-negative ABC that progressed during ET. Patients were enrolled between August 7, 2014, and December 29, 2015. Analyses for this report were conducted at the time of database lock on June 20, 2019.
Interventions: Patients were randomized 2:1 to receive abemaciclib or placebo, 150 mg, every 12 hours on a continuous schedule plus fulvestrant, 500 mg, per label. Randomization was stratified based on site of metastasis (visceral, bone only, or other) and resistance to prior ET (primary vs secondary).
Main outcomes and measures: The primary end point was investigator-assessed progression-free survival. Overall survival was a gated key secondary end point. The boundary P value for the interim analysis was .02.
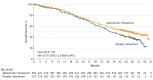
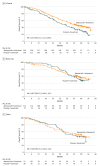
Results: Of 669 women enrolled, 446 (median [range] age, 59 [32-91] years) were randomized to the abemaciclib plus fulvestrant arm and 223 (median [range] age, 62 [32-87] years) were randomized to the placebo plus fulvestrant arm. At the prespecified interim, 338 deaths (77% of the planned 441 at the final analysis) were observed in the intent-to-treat population, with a median OS of 46.7 months for abemaciclib plus fulvestrant and 37.3 months for placebo plus fulvestrant (hazard ratio [HR], 0.757; 95% CI, 0.606-0.945; P = .01). Improvement in OS was consistent across all stratification factors. Among stratification factors, more pronounced effects were observed in patients with visceral disease (HR, 0.675; 95% CI, 0.511-0.891) and primary resistance to prior ET (HR, 0.686; 95% CI, 0.451-1.043). Time to second disease progression (median, 23.1 months vs 20.6 months), time to chemotherapy (median, 50.2 months vs 22.1 months), and chemotherapy-free survival (median, 25.5 months vs 18.2 months) were also statistically significantly improved in the abemaciclib arm vs placebo arm. No new safety signals were observed for abemaciclib.
Conclusions and relevance: Treatment with abemaciclib plus fulvestrant resulted in a statistically significant and clinically meaningful median OS improvement of 9.4 months for patients with HR-positive, ERBB2-negative ABC who progressed after prior ET regardless of menopausal status. Abemaciclib substantially delayed the receipt of subsequent chemotherapy.
Trial registration: ClinicalTrials.gov identifier: NCT02107703.
Conflict of interest statement
Figures



Comment in
-
CDK4/6 inhibitors prolong OS.Nat Rev Clin Oncol. 2019 Dec;16(12):722. doi: 10.1038/s41571-019-0288-z. Nat Rev Clin Oncol. 2019. PMID: 31605029 No abstract available.
-
Analysis of Overall Survival Benefit of Abemaciclib Plus Fulvestrant in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer.JAMA Oncol. 2020 Jul 1;6(7):1122. doi: 10.1001/jamaoncol.2020.1516. JAMA Oncol. 2020. PMID: 32463423 No abstract available.
-
Analysis of Overall Survival Benefit of Abemaciclib Plus Fulvestrant in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer.JAMA Oncol. 2020 Jul 1;6(7):1121-1122. doi: 10.1001/jamaoncol.2020.1513. JAMA Oncol. 2020. PMID: 32463424 No abstract available.
-
Analysis of Overall Survival Benefit of Abemaciclib Plus Fulvestrant in Hormone Receptor-Positive, ERBB2-Negative Breast Cancer-Reply.JAMA Oncol. 2020 Jul 1;6(7):1122-1123. doi: 10.1001/jamaoncol.2020.1518. JAMA Oncol. 2020. PMID: 32463426 No abstract available.
References
-
- Cristofanilli M, Turner NC, Bondarenko I, et al. . Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2016;17(4):425-439. doi:10.1016/S1470-2045(15)00613-0 - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

