Retention of Alzheimer Disease Research Participants
- PMID: 31567302
- PMCID: PMC6878201
- DOI: 10.1097/WAD.0000000000000353
Retention of Alzheimer Disease Research Participants
Abstract
Introduction: Participant retention is important to maintaining statistical power, minimizing bias, and preventing scientific error in Alzheimer disease and related dementias research.
Methods: We surveyed representative investigators from NIH-funded Alzheimer's Disease Research Centers (ADRC), querying their use of retention tactics across 12 strategies. We compared survey results to data from the National Alzheimer's Coordinating Center for each center. We used a generalized estimating equation with independent working covariance model and empirical standard errors to assess relationships between survey results and rates of retention, controlling for participant characteristics.
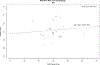
Results: Twenty-five (83%) responding ADRCs employed an average 42 (SD=7) retention tactics. In a multivariable model that accounted for participant characteristics, the number of retention tactics used by a center was associated with participant retention (odds ratio=1.68, 95% confidence interval: 1.42, 1.98; P<0.001 for the middle compared with the lowest tertile survey scores; odds ratio=1.59, 95% confidence interval: 1.30, 1.94; P<0.001 for the highest compared with the lowest tertile survey scores) at the first follow-up visit. Participant characteristics such as normal cognition diagnosis, older age, higher education, and Caucasian race were also associated with higher retention.
Conclusions: Retention in clinical research is more likely to be achieved by employing a variety of tactics.
Figures
References
-
- Kasenda B, von Elm E, You J, et al. Prevalence, characteristics, and publication of discontinued randomized trials. JAMA. 2014;311(10):1045–1051. - PubMed
-
- Fargo KN, Carrillo MC, Weiner MW, Potter WZ, Khachaturian Z. The crisis in recruitment for clinical trials in Alzheimer’s and dementia: An action plan for solutions. Alzheimers Dement. 2016;12(11):1113–1115. - PubMed
-
- Recruitment Vellas B., retention and other methodological issues related to clinical trials for Alzheimer’s disease. The journal of nutrition, health & aging. 2012;16(4):330. - PubMed
Publication types
MeSH terms
Grants and funding
- P50 AG016573/AG/NIA NIH HHS/United States
- P50 AG047266/AG/NIA NIH HHS/United States
- P30 AG010161/AG/NIA NIH HHS/United States
- P50 AG025688/AG/NIA NIH HHS/United States
- P50 AG005133/AG/NIA NIH HHS/United States
- UL1 TR000153/TR/NCATS NIH HHS/United States
- P50 AG047366/AG/NIA NIH HHS/United States
- P30 AG019610/AG/NIA NIH HHS/United States
- P30 AG028383/AG/NIA NIH HHS/United States
- P50 AG033514/AG/NIA NIH HHS/United States
- P30 AG013854/AG/NIA NIH HHS/United States
- P30 AG053760/AG/NIA NIH HHS/United States
- R21 AG056931/AG/NIA NIH HHS/United States
- P30 AG010124/AG/NIA NIH HHS/United States
- P50 AG023501/AG/NIA NIH HHS/United States
- R01 AG053555/AG/NIA NIH HHS/United States
- P50 AG005142/AG/NIA NIH HHS/United States
- P50 AG005131/AG/NIA NIH HHS/United States
- P30 AG010133/AG/NIA NIH HHS/United States
- P50 AG016574/AG/NIA NIH HHS/United States
- P50 AG005146/AG/NIA NIH HHS/United States
- U24 AG072122/AG/NIA NIH HHS/United States
- P30 AG035982/AG/NIA NIH HHS/United States
- P50 AG008702/AG/NIA NIH HHS/United States
- U01 AG016976/AG/NIA NIH HHS/United States
- P30 AG008051/AG/NIA NIH HHS/United States
- P50 AG005681/AG/NIA NIH HHS/United States
- P30 AG013846/AG/NIA NIH HHS/United States
- P50 AG047270/AG/NIA NIH HHS/United States
- P50 AG005136/AG/NIA NIH HHS/United States
- P30 AG072973/AG/NIA NIH HHS/United States
- P30 AG049638/AG/NIA NIH HHS/United States
- P30 AG012300/AG/NIA NIH HHS/United States
- P50 AG005134/AG/NIA NIH HHS/United States
- RF1 AG059407/AG/NIA NIH HHS/United States
- P30 AG008017/AG/NIA NIH HHS/United States
- UL1 TR001414/TR/NCATS NIH HHS/United States
- P50 AG005138/AG/NIA NIH HHS/United States
- P30 AG062715/AG/NIA NIH HHS/United States
- P30 AG010129/AG/NIA NIH HHS/United States

