The mechanism of thin filament regulation: Models in conflict?
- PMID: 31570503
- PMCID: PMC6829557
- DOI: 10.1085/jgp.201912446
The mechanism of thin filament regulation: Models in conflict?
Abstract
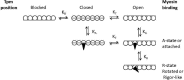
In a recent JGP article, Heeley et al. (2019. J. Gen. Physiol https://doi.org/10.1085/jgp.201812198) reopened the debate about two- versus three-state models of thin filament regulation. The authors review their work, which measures the rate constant of Pi release from myosin.ADP.Pi activated by actin or thin filaments under a variety of conditions. They conclude that their data can be described by a two-state model and raise doubts about the generally accepted three-state model as originally formulated by McKillop and Geeves (1993. Biophys. J. https://doi.org/10.1016/S0006-3495(93)81110-X). However, in the following article, we follow Plato's dictum that "twice and thrice over, as they say, good it is to repeat and review what is good." We have therefore reviewed the evidence for the three- and two-state models and present our view that the evidence is overwhelmingly in favor of three structural states of the thin filament, which regulate access of myosin to its binding sites on actin and, hence, muscle contractility.
© 2019 Geeves et al.
Figures
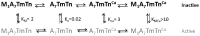
References
-
- Chalovich J.M., and Eisenberg E.. 1982. Inhibition of actomyosin ATPase activity by troponin-tropomyosin without blocking the binding of myosin to actin. J. Biol. Chem. 257:2432–2437 http://www.ncbi.nlm.nih.gov/pubmed/6460759. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

