Postzygotic inactivating mutations of RHOA cause a mosaic neuroectodermal syndrome
- PMID: 31570889
- PMCID: PMC6858542
- DOI: 10.1038/s41588-019-0498-4
Postzygotic inactivating mutations of RHOA cause a mosaic neuroectodermal syndrome
Erratum in
-
Author Correction: Postzygotic inactivating mutations of RHOA cause a mosaic neuroectodermal syndrome.Nat Genet. 2019 Nov;51(11):1660. doi: 10.1038/s41588-019-0527-3. Nat Genet. 2019. PMID: 31611689
-
Author Correction: Postzygotic inactivating mutations of RHOA cause a mosaic neuroectodermal syndrome.Nat Genet. 2020 Mar;52(3):353. doi: 10.1038/s41588-019-0565-x. Nat Genet. 2020. PMID: 32034319
Abstract
Hypopigmentation along Blaschko's lines is a hallmark of a poorly defined group of mosaic syndromes whose genetic causes are unknown. Here we show that postzygotic inactivating mutations of RHOA cause a neuroectodermal syndrome combining linear hypopigmentation, alopecia, apparently asymptomatic leukoencephalopathy, and facial, ocular, dental and acral anomalies. Our findings pave the way toward elucidating the etiology of pigmentary mosaicism and highlight the role of RHOA in human development and disease.
Conflict of interest statement
COMPETING FINANCIAL INTERESTS
The authors declare no competing financial interests.
Figures
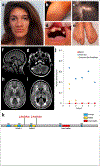

References
-
- Sybert VP Hypomelanosis of Ito: a description, not a diagnosis. J. Invest. Dermatol 103, S141–S143 (1994). - PubMed
-
- Pavone P, Praticò AD, Ruggieri M & Falsaperla R Hypomelanosis of Ito: a round on the frequency and type of epileptic complications. Neurol. Sci 36, 1173–1180 (2015). - PubMed
-
- Hall A Rho GTPases and the actin cytoskeleton. Science 279, 509–514 (1998). - PubMed
-
- Vetter IR & Wittinghofer A The guanine nucleotide-binding switch in three dimensions. Science 294, 1299–1304 (2001). - PubMed
Methods-only References
-
- Thevenon J et al. Diagnostic odyssey in severe neurodevelopmental disorders: toward clinical whole-exome sequencing as a first-line diagnostic test. Clin. Genet 89, 700–707 (2016). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

