Hypoxia-ischemia in the immature rodent brain impairs serotonergic neuronal function in certain dorsal raphé nuclei
- PMID: 31571657
- PMCID: PMC6921336
- DOI: 10.4103/1673-5374.266067
Hypoxia-ischemia in the immature rodent brain impairs serotonergic neuronal function in certain dorsal raphé nuclei
Abstract
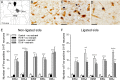
Neonatal hypoxia-ischemia (HI) results in losses of serotonergic neurons in specific dorsal raphé nuclei. However, not all serotonergic raphé neurons are lost and it is therefore important to assess the function of remaining neurons in order to understand their potential to contribute to neurological disorders in the HI-affected neonate. The main objective of this study was to determine how serotonergic neurons, remaining in the dorsal raphé nuclei after neonatal HI, respond to an external stimulus (restraint stress). On postnatal day 3 (P3), male rat pups were randomly allocated to one of the following groups: (i) control + no restraint (n = 5), (ii) control + restraint (n = 6), (iii) P3 HI + no restraint (n = 5) or (iv) P3 HI + restraint (n = 7). In the two HI groups, rat pups underwent surgery to ligate the common carotid artery and were then exposed to 6% O2 for 30 minutes. Six weeks after P3 HI, on P45, rats were subjected to restraint stress for 30 minutes. Using dual immunolabeling for Fos protein, a marker for neuronal activity, and serotonin (5-hydroxytrypamine; 5-HT), numbers of Fos-positive 5-HT neurons were determined in five dorsal raphé nuclei. We found that restraint stress alone increased numbers of Fos-positive 5-HT neurons in all five dorsal raphé nuclei compared to control animals. However, following P3 HI, the number of stress-induced Fos-positive 5-HT neurons was decreased significantly in the dorsal raphé ventrolateral, interfascicular and ventral nuclei compared with control animals exposed to restraint stress. In contrast, numbers of stress-induced Fos-positive 5-HT neurons in the dorsal raphé dorsal and caudal nuclei were not affected by P3 HI. These data indicate that not only are dorsal raphé serotonergic neurons lost after neonatal HI, but also remaining dorsal raphé serotonergic neurons have reduced differential functional viability in response to an external stimulus. Procedures were approved by the University of Queensland Animal Ethics Committee (UQCCR958/08/NHMRC) on February 27, 2009.
Keywords: Fos; dorsal raphénuclei; hypoxia-ischemia; neonate; newborn brain injury; preterm; restraint stress; serotonin.
Conflict of interest statement
None
Figures

Similar articles
-
Differential effects of neonatal hypoxic-ischemic brain injury on brainstem serotonergic raphe nuclei.Brain Res. 2010 Mar 31;1322:124-33. doi: 10.1016/j.brainres.2010.01.065. Epub 2010 Feb 1. Brain Res. 2010. PMID: 20122905
-
Neonatal hypoxia-ischaemia disrupts descending neural inputs to dorsal raphé nuclei.Neuroscience. 2013 Sep 17;248:427-35. doi: 10.1016/j.neuroscience.2013.06.016. Epub 2013 Jun 24. Neuroscience. 2013. PMID: 23806712
-
Disruption of raphé serotonergic neural projections to the cortex: a potential pathway contributing to remote loss of brainstem neurons following neonatal hypoxic-ischemic brain injury.Eur J Neurosci. 2012 Dec;36(11):3483-91. doi: 10.1111/j.1460-9568.2012.08276.x. Epub 2012 Sep 4. Eur J Neurosci. 2012. PMID: 22943572
-
The human raphe nuclei and the serotonergic system.J Chem Neuroanat. 2003 Dec;26(4):331-43. doi: 10.1016/j.jchemneu.2003.10.002. J Chem Neuroanat. 2003. PMID: 14729135 Review.
-
Functional topography of serotonergic systems supports the Deakin/Graeff hypothesis of anxiety and affective disorders.J Psychopharmacol. 2013 Dec;27(12):1090-106. doi: 10.1177/0269881113490328. Epub 2013 May 23. J Psychopharmacol. 2013. PMID: 23704363 Review.
References
-
- Abrams JK, Johnson PL, Hay-Schmidt A, Mikkelsen JD, Shekhar A, Lowry CA. Serotonergic systems associated with arousal and vigilance behaviors following administration of anxiogenic drugs. Neuroscience. 2005;133:983–997. - PubMed
-
- Anderson P, Doyle LW Victorian Infant Collaborative Study G. Neurobehavioral outcomes of school-age children born extremely low birth weight or very preterm in the 1990s. JAMA. 2003;289:3264–3272. - PubMed
-
- Anju TR, Korah PK, Jayanarayanan S, Paulose CS. Enhanced brain stem 5HT(2)A receptor function under neonatal hypoxic insult: role of glucose, oxygen, and epinephrine resuscitation. Mol Cell Biochem. 2011;354:151–160. - PubMed
-
- Baker KG, Halliday GM, Tork I. Cytoarchitecture of the human dorsal raphe nucleus. J Comp Neurol. 1990;301:147–161. - PubMed
LinkOut - more resources
Full Text Sources

