Investigation Of Vitamin B12-Modified Amphiphilic Sodium Alginate Derivatives For Enhancing The Oral Delivery Efficacy Of Peptide Drugs
- PMID: 31571874
- PMCID: PMC6760826
- DOI: 10.2147/IJN.S218944
Investigation Of Vitamin B12-Modified Amphiphilic Sodium Alginate Derivatives For Enhancing The Oral Delivery Efficacy Of Peptide Drugs
Abstract
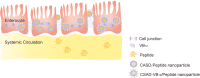
Purpose: Peptide drugs have been used in therapy various diseases. However, the poor bioavailability of peptide drugs for oral administration has limited their clinical applications, on account of the acidic environment and digestive enzymes inside the human gastrointestinal tract. To enhance stability in the human gastrointestinal tract, bioavailability, and targeted drug delivery of peptide drugs through oral administration, a vitamin B12-modified amphiphilic sodium alginate derivative (CSAD-VB12) was synthesized.
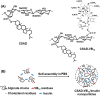
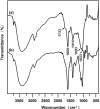
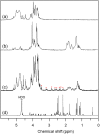
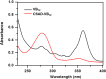
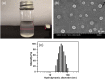
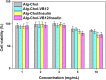
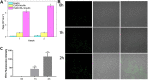
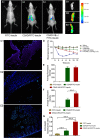
Materials and methods: A vitamin B12-modified amphiphilic sodium alginate derivative (CSAD-VB12) was synthesized via the N,N'-dicyclohexylcarbodiimide active method at room temperature, and then characterized using FTIR and 1H NMR spectroscopy. Insulin was used as a model peptide drug and the insulin-loaded CSAD-VB12 (CSAD-VB12/insulin) nanoparticles with negative zeta potentials were prepared in PBS (pH=7.4). Scanning electron microscopy was used to observe CSAD-VB12/insulin as spherical nanoparticles. The CSAD-VB12 derivatives and CSAD-VB12/insulin nanoparticles displayed nontoxicity towards the human colon adenocarcinoma (Caco-2) cells by CCK-8 test. Caco-2 cell model was used to measure the apparent permeability (Papp) of insulin, CSAD/insulin and CSAD-VB12/insulin. Furthermore, confocal was used to confirm the endocytosis of intestinal enterocytes. Type 1 diabetes mice were used to evaluate the intestinal absorption and retention effect of test nanoparticles.
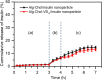
Results: They were observed as spherical nanoparticles in the size of 30-50 nm. The CSAD-VB12 derivatives and CSAD-VB12/insulin nanoparticles displayed nontoxicity towards the human colon adenocarcinoma (Caco-2) cells. Comparing with insulin and the CSAD/insulin nanoparticles, the CSAD-VB12/insulin nanoparticles exhibited higher permeation ability through intestinal enterocytes in the Caco-2 cell model. Oral administration of the CSAD-VB12/insulin nanoparticles to Type 1 diabetic mice yields higher intestinal retention effect, targeted absorption, and outstanding efficacy.
Conclusion: CSAD-VB12 derivatives enhance the small intestinal absorption efficacy and retention of peptide by oral administration, which indicated that it could be a promising candidate for oral peptide delivery in the prospective clinical application.
Keywords: human colon adenocarcinoma cell; insulin; nanoparticle; small intestinal absorption efficacy.
© 2019 Long et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

Similar articles
-
Enhancement of scutellarin oral delivery efficacy by vitamin B12-modified amphiphilic chitosan derivatives to treat type II diabetes induced-retinopathy.J Nanobiotechnology. 2017 Mar 1;15(1):18. doi: 10.1186/s12951-017-0251-z. J Nanobiotechnology. 2017. PMID: 28249594 Free PMC article.
-
Vitamin B12 functionalized layer by layer calcium phosphate nanoparticles: A mucoadhesive and pH responsive carrier for improved oral delivery of insulin.Acta Biomater. 2016 Feb;31:288-300. doi: 10.1016/j.actbio.2015.12.017. Epub 2015 Dec 11. Acta Biomater. 2016. PMID: 26685755
-
Efficient Peroral Delivery of Insulin via Vitamin B12 Modified Trimethyl Chitosan Nanoparticles.J Pharm Pharm Sci. 2015;18(2):155-70. doi: 10.18433/j3j88q. J Pharm Pharm Sci. 2015. PMID: 26158281
-
Recent Advances in Dietary Sources, Health Benefits, Emerging Encapsulation Methods, Food Fortification, and New Sensor-Based Monitoring of Vitamin B12: A Critical Review.Molecules. 2023 Nov 7;28(22):7469. doi: 10.3390/molecules28227469. Molecules. 2023. PMID: 38005191 Free PMC article. Review.
-
Oral delivery of anti-diabetes therapeutics using cell penetrating and transcytosing peptide strategies.Peptides. 2018 Feb;100:24-35. doi: 10.1016/j.peptides.2017.12.014. Peptides. 2018. PMID: 29412825 Review.
Cited by
-
Mucus interaction to improve gastrointestinal retention and pharmacokinetics of orally administered nano-drug delivery systems.J Nanobiotechnology. 2022 Aug 6;20(1):362. doi: 10.1186/s12951-022-01539-x. J Nanobiotechnology. 2022. PMID: 35933341 Free PMC article. Review.
-
Bile acid transporter-mediated oral drug delivery.J Control Release. 2020 Nov 10;327:100-116. doi: 10.1016/j.jconrel.2020.07.034. Epub 2020 Jul 22. J Control Release. 2020. PMID: 32711025 Free PMC article. Review.
-
Emerging trends of biomedical nanotechnology in nutrition, health monitoring and disease diagnosis.3 Biotech. 2025 Jun;15(6):152. doi: 10.1007/s13205-025-04291-9. Epub 2025 May 5. 3 Biotech. 2025. PMID: 40336812 Review.
-
Mucoadhesive carriers for oral drug delivery.J Control Release. 2022 Nov;351:504-559. doi: 10.1016/j.jconrel.2022.09.024. Epub 2022 Sep 30. J Control Release. 2022. PMID: 36116580 Free PMC article. Review.
-
Cellular Uptake and Transport Mechanism of 6-Mercaptopurine Nanomedicines for Enhanced Oral Bioavailability.Int J Nanomedicine. 2023 Jan 5;18:79-94. doi: 10.2147/IJN.S394819. eCollection 2023. Int J Nanomedicine. 2023. PMID: 36636639 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

