miR-361 enhances sensitivity to 5-fluorouracil by targeting the FOXM1-ABCC5/10 signaling pathway in colorectal cancer
- PMID: 31579069
- PMCID: PMC6757261
- DOI: 10.3892/ol.2019.10741
miR-361 enhances sensitivity to 5-fluorouracil by targeting the FOXM1-ABCC5/10 signaling pathway in colorectal cancer
Abstract
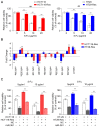
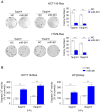
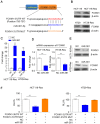
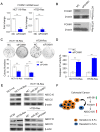
Colorectal cancer (CRC) is one of most common malignancies worldwide. 5-fluorouracil (5-FU) is a mainstay of CRC treatment, particularly in patients with advanced stages of the disease; however, 5-FU-based chemotherapy is not always effective and may result in progression of the disease. The present study investigated several candidate microRNAs (miRs) in parental and 5-FU-resistant HCT116 and HT29 cells, and identified miR-361 as a novel regulator of chemosensitivity. Overexpression of miR-361 enhanced the 5-FU susceptibility of parental and resistant HCT116 and HT29 cells in vitro. Impaired colony formation capacity and increased cell apoptosis (as determined via flow cytometry) was observed in resistant HCT116 and HT29 cells. Furthermore, forkhead box M1 (FOXM1) was identified as a target gene of miR-361 using a dual-luciferase reporter assay, western blotting and reverse transcription-quantitative PCR. Additionally, FOXM1 knockdown improved the cytotoxicity of 5-FU in resistant CRC. ATP binding cassette subfamily C members 5 and 10 (ABCC5/10) were found to be downstream effectors of miR-361. In conclusion, miR-361 increased chemosensitivity, at least in part, via modulation of FOXM1-ABCC5/10. miR-361 may serve as a potential therapeutic target for patients with CRC.
Keywords: 5-fluorouracil; ATP binding cassette subfamily C; chemosensitivity; colorectal cancer; forkhead box M1; microRNA-361.
Copyright: © Zhang et al.
Figures

Similar articles
-
MicroRNA-149 Increases the Sensitivity of Colorectal Cancer Cells to 5-Fluorouracil by Targeting Forkhead Box Transcription Factor FOXM1.Cell Physiol Biochem. 2016;39(2):617-29. doi: 10.1159/000445653. Epub 2016 Jul 15. Cell Physiol Biochem. 2016. PMID: 27415661
-
Circ_0007031 enhances tumor progression and promotes 5-fluorouracil resistance in colorectal cancer through regulating miR-133b/ABCC5 axis.Cancer Biomark. 2020;29(4):531-542. doi: 10.3233/CBM-200023. Cancer Biomark. 2020. PMID: 32865180
-
Circ-PRKDC Contributes to 5-Fluorouracil Resistance of Colorectal Cancer Cells by Regulating miR-375/FOXM1 Axis and Wnt/β-Catenin Pathway.Onco Targets Ther. 2020 Jun 23;13:5939-5953. doi: 10.2147/OTT.S253468. eCollection 2020. Onco Targets Ther. 2020. PMID: 32606803 Free PMC article.
-
MicroRNA-23a antisense enhances 5-fluorouracil chemosensitivity through APAF-1/caspase-9 apoptotic pathway in colorectal cancer cells.J Cell Biochem. 2014 Apr;115(4):772-84. doi: 10.1002/jcb.24721. J Cell Biochem. 2014. PMID: 24249161
-
FOXM1 evokes 5-fluorouracil resistance in colorectal cancer depending on ABCC10.Oncotarget. 2017 Jan 31;8(5):8574-8589. doi: 10.18632/oncotarget.14351. Oncotarget. 2017. PMID: 28051999 Free PMC article.
Cited by
-
5-Fluorouracil: A Narrative Review on the Role of Regulatory Mechanisms in Driving Resistance to This Chemotherapeutic Agent.Front Oncol. 2021 Apr 19;11:658636. doi: 10.3389/fonc.2021.658636. eCollection 2021. Front Oncol. 2021. PMID: 33954114 Free PMC article. Review.
-
Downregulation of miR-199a-3p in Hepatocellular Carcinoma and Its Relevant Molecular Mechanism via GEO, TCGA Database and In Silico Analyses.Technol Cancer Res Treat. 2020 Jan-Dec;19:1533033820979670. doi: 10.1177/1533033820979670. Technol Cancer Res Treat. 2020. PMID: 33327879 Free PMC article.
-
Elucidation of underlying molecular mechanism of 5-Fluorouracil chemoresistance and its restoration using fish oil in experimental colon carcinoma.Mol Cell Biochem. 2021 Mar;476(3):1517-1527. doi: 10.1007/s11010-020-03999-x. Epub 2021 Jan 4. Mol Cell Biochem. 2021. PMID: 33392922
-
Effect of TTLL12 on tubulin tyrosine nitration as a novel target for screening anticancer drugs in vitro.Oncol Lett. 2020 Dec;20(6):340. doi: 10.3892/ol.2020.12203. Epub 2020 Oct 8. Oncol Lett. 2020. PMID: 33123251 Free PMC article.
-
Progress and challenges of multidrug resistance proteins in diseases.Am J Cancer Res. 2022 Oct 15;12(10):4483-4501. eCollection 2022. Am J Cancer Res. 2022. PMID: 36381332 Free PMC article. Review.
References
-
- Montagnani F, Chiriatti A, Turrisi G, Francini G, Fiorentini G. A systematic review of FOLFOXIRI chemotherapy for the first-line treatment of metastatic colorectal cancer: Improved efficacy at the cost of increased toxicity. Colorectal Dis. 2011;13:846–852. doi: 10.1111/j.1463-1318.2010.02206.x. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
