Delivery of RIPK4 small interfering RNA for bladder cancer therapy using natural halloysite nanotubes
- PMID: 31579820
- PMCID: PMC6760933
- DOI: 10.1126/sciadv.aaw6499
Delivery of RIPK4 small interfering RNA for bladder cancer therapy using natural halloysite nanotubes
Abstract
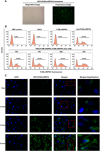
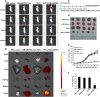
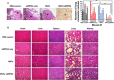
RNA interference (RNAi) technology can specifically silence the expression of a target gene and has emerged as a promising therapeutic method to treat cancer. In the present study, we showed that natural halloysite nanotube (HNT)-assisted delivery of an active small interfering RNA (siRNA) targeting receptor-interacting protein kinase 4 ( RIPK4 ) efficiently silenced its expression to treat bladder cancer. The HNTs/siRNA complex increased the serum stability of the siRNA, increased its circulation lifetime in blood, and promoted the cellular uptake and tumor accumulation of the siRNA. The siRNA markedly down-regulated RIPK4 expression in bladder cancer cells and bladder tumors, thus inhibiting tumorigenesis and progression in three bladder tumor models (a subcutaneous model, an in situ bladder tumor model, and a lung metastasis model), with no adverse effects. Thus, we revealed a simple but effective method to inhibit bladder cancer using RIPK4 silencing, indicating a promising therapeutic method for bladder cancer.
Copyright © 2019 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
Figures



Similar articles
-
Fe-doped chrysotile nanotubes containing siRNAs to silence SPAG5 to treat bladder cancer.J Nanobiotechnology. 2021 Jun 23;19(1):189. doi: 10.1186/s12951-021-00935-z. J Nanobiotechnology. 2021. PMID: 34162370 Free PMC article.
-
Multifunctional nanocarrier based on clay nanotubes for efficient intracellular siRNA delivery and gene silencing.J Biomater Appl. 2014 Apr;28(8):1180-9. doi: 10.1177/0885328213501215. Epub 2013 Aug 28. J Biomater Appl. 2014. PMID: 23985535
-
RNAi-based therapeutics targeting survivin and PLK1 for treatment of bladder cancer.Mol Ther. 2011 May;19(5):928-35. doi: 10.1038/mt.2011.21. Epub 2011 Mar 1. Mol Ther. 2011. PMID: 21364537 Free PMC article.
-
The design and exogenous delivery of siRNA for post-transcriptional gene silencing.J Drug Target. 2004 Jul;12(6):315-40. doi: 10.1080/10611860400006257. J Drug Target. 2004. PMID: 15545082 Review.
-
The potential and advances in RNAi therapy: chemical and structural modifications of siRNA molecules and use of biocompatible nanocarriers.J Control Release. 2014 Nov 10;193:113-21. doi: 10.1016/j.jconrel.2014.05.030. Epub 2014 May 24. J Control Release. 2014. PMID: 24862319 Review.
Cited by
-
Depletion of RIPK4 parallels higher malignancy potential in cutaneous squamous cell carcinoma.PeerJ. 2022 Feb 10;10:e12932. doi: 10.7717/peerj.12932. eCollection 2022. PeerJ. 2022. PMID: 35186499 Free PMC article.
-
CRISPR-Cas9 potential for identifying novel therapeutic targets in muscle-invasive bladder cancer.Nat Rev Urol. 2025 Jan;22(1):55-65. doi: 10.1038/s41585-024-00901-y. Epub 2024 Jul 1. Nat Rev Urol. 2025. PMID: 38951705 Review.
-
RBPMS inhibits bladder cancer metastasis by downregulating MYC pathway through alternative splicing of ANKRD10.Commun Biol. 2025 Mar 5;8(1):367. doi: 10.1038/s42003-025-07842-1. Commun Biol. 2025. PMID: 40044952 Free PMC article.
-
Current advance of nanotechnology in diagnosis and treatment for malignant tumors.Signal Transduct Target Ther. 2024 Aug 12;9(1):200. doi: 10.1038/s41392-024-01889-y. Signal Transduct Target Ther. 2024. PMID: 39128942 Free PMC article. Review.
-
Insight Into the Function of RIPK4 in Keratinocyte Differentiation and Carcinogenesis.Front Oncol. 2020 Aug 14;10:1562. doi: 10.3389/fonc.2020.01562. eCollection 2020. Front Oncol. 2020. PMID: 32923402 Free PMC article. Review.
References
-
- Grimm D., Small silencing RNAs: State-of-the-art. Adv. Drug Deliv. Rev. 61, 672–703 (2009). - PubMed
-
- Matsumura Y., Maeda H., A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 46, 6387–6392 (1986). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

