Synovial tissue signatures enhance clinical classification and prognostic/treatment response algorithms in early inflammatory arthritis and predict requirement for subsequent biological therapy: results from the pathobiology of early arthritis cohort (PEAC)
- PMID: 31582377
- PMCID: PMC6900253
- DOI: 10.1136/annrheumdis-2019-215751
Synovial tissue signatures enhance clinical classification and prognostic/treatment response algorithms in early inflammatory arthritis and predict requirement for subsequent biological therapy: results from the pathobiology of early arthritis cohort (PEAC)
Abstract
Objective: To establish whether synovial pathobiology improves current clinical classification and prognostic algorithms in early inflammatory arthritis and identify predictors of subsequent biological therapy requirement.
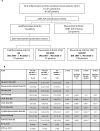
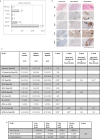
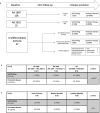
Methods: 200 treatment-naïve patients with early arthritis were classified as fulfilling RA1987 American College of Rheumatology (ACR) criteria (RA1987) or as undifferentiated arthritis (UA) and patients with UA further classified into those fulfilling RA2010 ACR/European League Against Rheumatism (EULAR) criteria. Treatment requirements at 12 months (Conventional Synthetic Disease Modifying Antirheumatic Drugs (csDMARDs) vs biologics vs no-csDMARDs treatment) were determined. Synovial tissue was retrieved by minimally invasive, ultrasound-guided biopsy and underwent processing for immunohistochemical (IHC) and molecular characterisation. Samples were analysed for macrophage, plasma-cell and B-cells and T-cells markers, pathotype classification (lympho-myeloid, diffuse-myeloid or pauci-immune) by IHC and gene expression profiling by Nanostring.
Results: 128/200 patients were classified as RA1987, 25 as RA2010 and 47 as UA. Patients classified as RA1987 criteria had significantly higher levels of disease activity, histological synovitis, degree of immune cell infiltration and differential upregulation of genes involved in B and T cell activation/function compared with RA2010 or UA, which shared similar clinical and pathobiological features. At 12-month follow-up, a significantly higher proportion of patients classified as lympho-myeloid pathotype required biological therapy. Performance of a clinical prediction model for biological therapy requirement was improved by the integration of synovial pathobiological markers from 78.8% to 89%-90%.
Conclusion: The capacity to refine early clinical classification criteria through synovial pathobiological markers offers the potential to predict disease outcome and stratify therapeutic intervention to patients most in need.
Keywords: Early Rheumatoid Arthritis; Synovitis; Ultrasonography.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

