Cyclic Changes in Active Site Polarization and Dynamics Drive the 'Ping-pong' Kinetics in NRH:Quinone Oxidoreductase 2: An Insight from QM/MM Simulations
- PMID: 31583178
- PMCID: PMC6776251
- DOI: 10.1021/acscatal.8b04193
Cyclic Changes in Active Site Polarization and Dynamics Drive the 'Ping-pong' Kinetics in NRH:Quinone Oxidoreductase 2: An Insight from QM/MM Simulations
Abstract
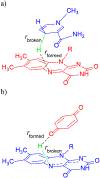
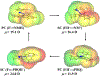
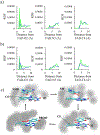
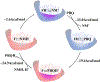
Quinone reductases belong to the family of flavin-dependent oxidoreductases. With the redox active cofactor, flavin adenine dinucleotide, quinone reductases are known to utilize a 'ping-pong' kinetic mechanism during catalysis in which a hydride is bounced back and forth between flavin and its two substrates. However, the continuation of this catalytic cycle requires product displacement steps, where the product of one redox half-cycle is displaced by the substrate of the next half-cycle. Using improved hybrid quantum mechanical/molecular mechanical simulations, both the catalytic hydride transfer and the product displacement reactions were studied in NRH:quinone oxidoreductase 2. Initially, the self-consistent charge-density functional tight binding theory was used to describe flavin ring and the substrate atoms, while embedded in the molecular mechanically-treated solvated active site. Then, for each step of the catalytic cycle, a further improvement of energetics was made using density functional theory-based corrections. The present study showcases an integrated interplay of solvation, protonation, and protein matrix-induced polarization as the driving force behind the thermodynamic wheel of the 'ping-pong' kinetics. Reported here is the first-principles model of the 'ping-pong' kinetics that portrays how cyclic changes in the active site polarization and dynamics govern the oscillatory hydride transfer and product displacement in this enzyme.
Keywords: Double displacement reactions; Kohn-Sham density functional theory; flavoenzyme; hydride transfer reactions; quantum mechanical/molecular mechanical calculations; quinone oxidoreductase 2; self-consistent charge density functional tight-binding theory; ‘ping-pong’ kinetics.
Figures


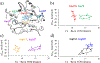


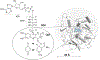

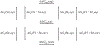
References
-
- Sollner S; Macheroux P, New Roles of Flavoproteins in Molecular Cell Biology: An Unexpected Role for Quinone Reductases as Regulators of Proteasomal Degradation. FEBS J. 2009, 276, 4313–4324. - PubMed
-
- Santina E The Potential Role of the Oxidoreductase, NQO2 in Breast Cancer. Doctoral dissertation, University of Manchester, Manchester, September 2015.
-
- Boutin JA, Quinone Reductase 2 as a Promising Target of Melatonin Therapeutic Actions. Expert Opin. Ther. Targets 2016, 20, 303–317. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
