Molecular mechanisms of heart regeneration
- PMID: 31587963
- PMCID: PMC7071978
- DOI: 10.1016/j.semcdb.2019.09.003
Molecular mechanisms of heart regeneration
Abstract
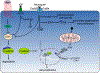
The adult mammalian heart is incapable of clinically relevant regeneration. The regenerative deficit in adult mammalian heart contrasts with the fetal and neonatal heart, which demonstrate substantial regenerative capacity after injury. This deficiency in adult mammals is attributable to the lack of resident stem cells after birth, combined with an inability of pre-existing cardiomyocytes to complete cytokinesis. Studies of neonatal heart regeneration in mammals suggest that latent regenerative potential can be re-activated. Dissecting the cellular and molecular mechanisms that promote cardiomyocyte proliferation is key to stimulating true regeneration in adult humans. Here, we review recent advances in our understanding of cardiomyocyte proliferation that suggest molecular approaches to heart regeneration.
Keywords: Cardiomyocyte proliferation; Growth factors; Heart regeneration; Hippo-YAP; Wnt.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Figures


References
-
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O'Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS, E. American Heart Association Council on, C. Prevention Statistics, S. Stroke Statistics, Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association, Circulation 139(10) (2019) e56–e528. - PubMed
-
- van den Borne SW, Diez J, Blankesteijn WM, Verjans J, Hofstra L, Narula J, Myocardial remodeling after infarction: the role of myofibroblasts, Nat Rev Cardiol 7(1) (2010) 30–7. - PubMed
-
- Whelan RS, Kaplinskiy V, Kitsis RN, Cell death in the pathogenesis of heart disease: mechanisms and significance, Annu Rev Physiol 72 (2010) 19–44 - PubMed
-
- Verheugt FW, Gersh BJ, Armstrong PW, Aborted myocardial infarction: a new target for reperfusion therapy, Eur Heart J 27(8) (2006) 901–4. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

