Hepatic steatosis in patients with acromegaly
- PMID: 31592448
- PMCID: PMC6775446
- DOI: 10.1002/edm2.90
Hepatic steatosis in patients with acromegaly
Abstract
Objective: Comorbid NAFLD is increasingly being diagnosed in patients with diabetes and nondiabetic endocrinopathies. The aim of this study was to assess hepatic steatosis noninvasively by transient elastography in patients with acromegaly.
Design: A cross-sectional study including 22 patients with acromegaly.
Methods: Hepatic steatosis was quantified using controlled attenuation parameter (CAP) during elastography. Anthropometric measurements were obtained, serum liver function tests and lipid and hormone profiles were measured, and prosteatogenic gene variants were genotyped using standard assays.
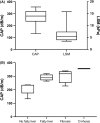
Results: In total, 41% of patients were women (mean age 60 ± 14.7 years, mean BMI 31.2 ± 4.6 kg/m2). Hepatic steatosis, as defined by CAP > 248 dB/m, was present in 66% of patients. Five (45%) of the patients with hepatic steatosis also had fibrosis, and one presented with cirrhosis. Nine patients were carriers of the PNPLA3 p.I148M prosteatogenic [M] risk allele, eight of whom were heterozygotes. CAP values were significantly (P = .045) higher in these patients and corresponded to advanced steatosis, as compared to patients with the wild-type genotype, who demonstrated CAP values consistent with mild steatosis (311 ± 33 dB/m. vs 254 ± 62 dB/m). CAP values did not differ significantly in carriers of distinct TM6SF2 and MBOAT7 genotypes; however, carriers of the risk alleles displayed higher CAP as compared to wild-type patients.
Conclusions: This study shows that in patients with acromegaly, carriers of the PNPLA3 susceptibility allele are at risk of developing hepatic steatosis, as assessed by CAP. Comorbid NAFLD might compound prognosis in such patients; thus, further research into the pathomechanisms and treatment of NAFLD in acromegaly is warranted.
Keywords: MBOAT7; TM6SF2; adiponutrin; controlled attenuation parameter; nonalcoholic fatty liver disease; transient elastography.
© 2019 The Authors. Endocrinology, Diabetes & Metabolism published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors of this study explicitly declare that there are no conflicts of interest in connection with this article.
Figures

References
-
- Stefan N, Haring HU, Cusi K. Non‐alcoholic fatty liver disease: causes, diagnosis, cardiometabolic consequences, and treatment strategies. Lancet Diabetes Endocrinol. 2018;7(4):313‐324. - PubMed
-
- Pingitore P, Romeo S. The role of PNPLA3 in health and disease. Biochim Biophys Acta. 2018;1864(6):900‐906. - PubMed
-
- Eslam M, Valenti L, Romeo S. Genetics and epigenetics of NAFLD and NASH: Clinical impact. J Hepatol. 2018;68(2):268‐279. - PubMed
-
- Arab JP, Arrese M, Trauner M. Recent insights into the pathogenesis of nonalcoholic fatty liver disease. Annu Rev Pathol. 2018;13:321‐350. - PubMed
-
- Targher G, Lonardo A, Byrne CD. Nonalcoholic fatty liver disease and chronic vascular complications of diabetes mellitus. Nat Rev Endocrinol. 2018;14(2):99‐114. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

