Recalibration of the insect evolutionary time scale using Monte San Giorgio fossils suggests survival of key lineages through the End-Permian Extinction
- PMID: 31594499
- PMCID: PMC6790769
- DOI: 10.1098/rspb.2019.1854
Recalibration of the insect evolutionary time scale using Monte San Giorgio fossils suggests survival of key lineages through the End-Permian Extinction
Abstract
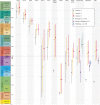
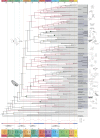
Insects are a highly diverse group of organisms and constitute more than half of all known animal species. They have evolved an extraordinary range of traits, from flight and complete metamorphosis to complex polyphenisms and advanced eusociality. Although the rich insect fossil record has helped to chart the appearance of many phenotypic innovations, data are scarce for a number of key periods. One such period is that following the End-Permian Extinction, recognized as the most catastrophic of all extinction events. We recently discovered several 240-million-year-old insect fossils in the Mount San Giorgio Lagerstätte (Switzerland-Italy) that are remarkable for their state of preservation (including internal organs and soft tissues), and because they extend the records of their respective taxa by up to 200 million years. By using these fossils as calibrations in a phylogenomic dating analysis, we present a revised time scale for insect evolution. Our date estimates for several major lineages, including the hyperdiverse crown groups of Lepidoptera, Hemiptera: Heteroptera and Diptera, are substantially older than their currently accepted post-Permian origins. We found that major evolutionary innovations, including flight and metamorphosis, appeared considerably earlier than previously thought. These results have numerous implications for understanding the evolution of insects and their resilience in the face of extreme events such as the End-Permian Extinction.
Keywords: divergence times; fossil calibration; molecular dating; phylogenomics; relaxed molecular clock.
Conflict of interest statement
We declare we have no competing interests
Figures

References
-
- Grimaldi D, Engel MS. 2005. Evolution of the insects. Cambridge, UK: Cambridge University Press.
