Structure-Based Design, Synthesis and Biological Evaluation of Bis-Tetrahydropyran Furan Acetogenin Mimics Targeting the Trypanosomatid F1 Component of ATP Synthase
- PMID: 31598093
- PMCID: PMC6774295
- DOI: 10.1002/ejoc.201900541
Structure-Based Design, Synthesis and Biological Evaluation of Bis-Tetrahydropyran Furan Acetogenin Mimics Targeting the Trypanosomatid F1 Component of ATP Synthase
Abstract
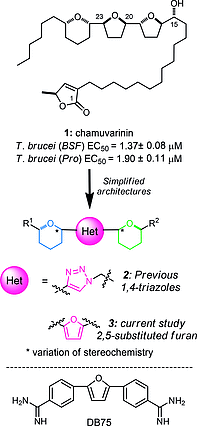
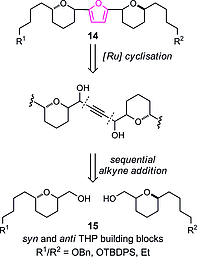
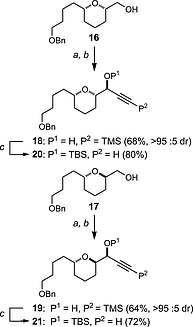
The protozoan parasites Trypanosoma brucei, Trypanosoma cruzi and Leishmania spp. are responsible for the severely debilitating neglected Tropical diseases of African sleeping sickness, Chagas disease and leishmaniasis, respectively. As part of our ongoing programme exploring the potential of simplified analogues of the acetogenin chamuvarinin we identified the T. brucei FoF1-ATP synthase as a target of our earlier triazole analogue series. Using computational docking studies, we hypothesised that the central triazole heterocyclic spacer could be substituted for a central 2,5-substituted furan moiety, thus diversifying the chemical framework for the generation of compounds with greater potency and/or selectivity. Here we report the design, docking, synthesis and biological evaluation of new series of trypanocidal compounds and demonstrate their on-target inhibitory effects. Furthermore, the synthesis of furans by the modular coupling of alkyne- and aldehyde-THPs to bis-THP 1,4-alkyne diols followed by ruthenium/xantphos-catalysed heterocyclisation described here represents the most complex use of this method of heterocyclisation to date.
Keywords: Biological activity; Drug design; FoF1‐ATP synthase; Homogeneous catalysis; Trypanosomatid.
© 2019 The Authors. Published by Wiley‐VCH Verlag GmbH & Co. KGaA.
Figures


Similar articles
-
Photo-affinity labelling and biochemical analyses identify the target of trypanocidal simplified natural product analogues.PLoS Negl Trop Dis. 2017 Sep 5;11(9):e0005886. doi: 10.1371/journal.pntd.0005886. eCollection 2017 Sep. PLoS Negl Trop Dis. 2017. PMID: 28873407 Free PMC article.
-
Simplifying nature: Towards the design of broad spectrum kinetoplastid inhibitors, inspired by acetogenins.Bioorg Med Chem. 2017 Nov 15;25(22):6126-6136. doi: 10.1016/j.bmc.2017.01.021. Epub 2017 Jan 28. Bioorg Med Chem. 2017. PMID: 28185724
-
Non-natural acetogenin analogues as potent Trypanosoma brucei inhibitors.ChemMedChem. 2014 Nov;9(11):2548-56. doi: 10.1002/cmdc.201402272. Epub 2014 Aug 21. ChemMedChem. 2014. PMID: 25145275 Free PMC article.
-
Exploring N-myristoyltransferase as a promising drug target against parasitic neglected tropical diseases.Eur J Med Chem. 2023 Oct 5;258:115550. doi: 10.1016/j.ejmech.2023.115550. Epub 2023 Jun 8. Eur J Med Chem. 2023. PMID: 37336067 Review.
-
The use of Sulfonamide Derivatives in the Treatment of Trypanosomatid Parasites including Trypanosoma cruzi, Trypanosoma brucei, and Leishmania ssp.Med Chem. 2020;16(1):24-38. doi: 10.2174/1573406415666190620141109. Med Chem. 2020. PMID: 31218962 Review.
Cited by
-
QSAR Modeling for Multi-Target Drug Discovery: Designing Simultaneous Inhibitors of Proteins in Diverse Pathogenic Parasites.Front Chem. 2021 Mar 10;9:634663. doi: 10.3389/fchem.2021.634663. eCollection 2021. Front Chem. 2021. PMID: 33777898 Free PMC article.
References
-
- World Health Organization, “Trypanosomiasis, African,” 2018.
-
- World Health Organization, “Leishmaniasis,” 2018.
-
- Hirth B., Barker R. H., Celatka C. a., Klinger J. D., Liu H., Nare B., Nijjar A., Phillips M. a., Sybertz E., Willert E. K., et al., Bioorg. Med. Chem. Lett., 2009, 19, 2916–2919. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
