Histology Atlas of the Developing Mouse Urinary System With Emphasis on Prenatal Days E10.5-E18.5
- PMID: 31599209
- PMCID: PMC6814567
- DOI: 10.1177/0192623319873871
Histology Atlas of the Developing Mouse Urinary System With Emphasis on Prenatal Days E10.5-E18.5
Abstract
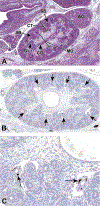
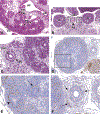
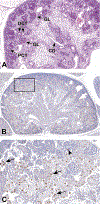
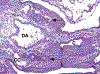
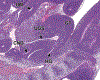
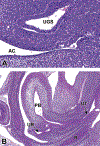
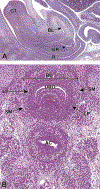
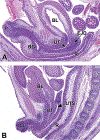
Congenital abnormalities of the urinary tract are some of the most common human developmental abnormalities. Several genetically engineered mouse models have been developed to mimic these abnormalities and aim to better understand the molecular mechanisms of disease. This atlas has been developed as an aid to pathologists and other biomedical scientists for identification of abnormalities in the developing murine urinary tract by cataloguing normal structures at each stage of development. Hematoxylin and eosin- and immunohistochemical-stained sections are provided, with a focus on E10.5-E18.5, as well as a brief discussion of postnatal events in urinary tract development. A section on abnormalities in the development of the urinary tract is also provided, and molecular mechanisms are presented as supplementary material. Additionally, overviews of the 2 key processes of kidney development, branching morphogenesis and nephrogenesis, are provided to aid in the understanding of the complex organogenesis of the kidney. One of the key findings of this atlas is the histological identification of the ureteric bud at E10.5, as previous literature has provided conflicting reports on the initial point of budding. Furthermore, attention is paid to points where murine development is significantly distinct from human development, namely, in the cessation of nephrogenesis.
Keywords: atlas; bladder; development; embryo; genetically engineered mice; kidney; urethra.
Conflict of interest statement
Declaration of Conflicting Interests Statement
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures





References
-
- Pohl M, Bhatnagar V, Mendoza SA, Nigam SK. Toward an etiological classification of developmental disorders of the kidney and upper urinary tract. Kidney Int 2002;61(1):10–19. - PubMed
-
- North American Pediatric Renal Trials and Collaborative Studies. 2008 Annual Report 2008.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

