Intratumoral delivery of RIG-I agonist SLR14 induces robust antitumor responses
- PMID: 31601678
- PMCID: PMC6888973
- DOI: 10.1084/jem.20190801
Intratumoral delivery of RIG-I agonist SLR14 induces robust antitumor responses
Abstract
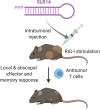
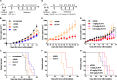
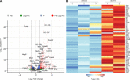
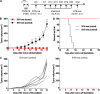
Cytosolic nucleic acid-sensing pathways can be triggered to enhance immune response to cancer. In this study, we tested the antitumor activity of a unique RIG-I agonist, stem loop RNA (SLR) 14. In the immunogenic tumor models, we observed significant tumor growth delay and an extended survival in SLR14-treated mice. SLR14 also greatly improved antitumor efficacy of anti-PD1 antibody over single-agent treatment. SLR14 was mainly taken up by CD11b+ myeloid cells in the tumor microenvironment, and many genes associated with immune defense were significantly up-regulated after treatment, accompanied by increase in the number of CD8+ T lymphocytes, NK cells, and CD11b+ cells in SLR14-treated tumors. Strikingly, SLR14 dramatically inhibited nonimmunogenic B16 tumor growth, and the cured mice developed an immune memory. Furthermore, a systemic antitumor response was observed in both bilateral and tumor metastasis models. Collectively, our results demonstrate that SLR14 is a promising therapeutic RIG-I agonist for cancer treatment, either alone or in combination with existing immunotherapies.
© 2019 Jiang et al.
Figures







References
-
- Besch R., Poeck H., Hohenauer T., Senft D., Häcker G., Berking C., Hornung V., Endres S., Ruzicka T., Rothenfusser S., and Hartmann G.. 2009. Proapoptotic signaling induced by RIG-I and MDA-5 results in type I interferon-independent apoptosis in human melanoma cells. J. Clin. Invest. 119:2399–2411. - PMC - PubMed
-
- Binnewies M., Roberts E.W., Kersten K., Chan V., Fearon D.F., Merad M., Coussens L.M., Gabrilovich D.I., Ostrand-Rosenberg S., Hedrick C.C., et al. . 2018. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 24:541–550. 10.1038/s41591-018-0014-x - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

