Garnet, the archetypal cubic mineral, grows tetragonal
- PMID: 31605020
- PMCID: PMC6789019
- DOI: 10.1038/s41598-019-51214-9
Garnet, the archetypal cubic mineral, grows tetragonal
Abstract
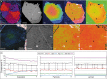
Garnet is the archetypal cubic mineral, occurring in a wide variety of rock types in Earth's crust and upper mantle. Owing to its prevalence, durability and compositional diversity, garnet is used to investigate a broad range of geological processes. Although birefringence is a characteristic feature of rare Ca-Fe3+ garnet and Ca-rich hydrous garnet, the optical anisotropy that has occasionally been documented in common (that is, anhydrous Ca-Fe2+-Mg-Mn) garnet is generally attributed to internal strain of the cubic structure. Here we show that common garnet with a non-cubic (tetragonal) crystal structure is much more widespread than previously thought, occurring in low-temperature, high-pressure metamorphosed basalts (blueschists) from subduction zones and in low-grade metamorphosed mudstones (phyllites and schists) from orogenic belts. Indeed, a non-cubic symmetry appears to be typical of common garnet that forms at low temperatures (<450 °C), where it has a characteristic Fe-Ca-rich composition with very low Mg contents. We propose that, in most cases, garnet does not initially grow cubic. Our discovery indicates that the crystal chemistry and thermodynamic properties of garnet at low-temperature need to be re-assessed, with potential consequences for the application of garnet as an investigative tool in a broad range of geological environments.
Conflict of interest statement
The authors declare no competing interests.
Figures







References
LinkOut - more resources
Full Text Sources

