Neural Interactions in a Spatially-Distributed Cortical Network During Perceptual Decision-Making
- PMID: 31607873
- PMCID: PMC6769171
- DOI: 10.3389/fnbeh.2019.00220
Neural Interactions in a Spatially-Distributed Cortical Network During Perceptual Decision-Making
Abstract
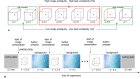
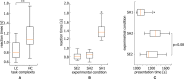
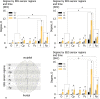
Behavioral experiments evidence that attention is not maintained at a constant level, but fluctuates with time. Recent studies associate such fluctuations with dynamics of attention-related cortical networks, however the exact mechanism remains unclear. To address this issue, we consider functional neuronal interactions during the accomplishment of a reaction time (RT) task which requires sustained attention. The participants are subjected to a binary classification of a large number of presented ambiguous visual stimuli with different degrees of ambiguity. Generally, high ambiguity causes high RT and vice versa. However, we demonstrate that RT fluctuates even when the stimulus ambiguity remains unchanged. The analysis of neuronal activity reveals that the subject's behavioral response is preceded by the formation of a distributed functional network in the β-frequency band. This network is characterized by high connectivity in the frontal cortex and supposed to subserve a decision-making process. We show that neither the network structure nor the duration of its formation depend on RT and stimulus ambiguity. In turn, RT is related to the moment of time when the β-band functional network emerges. We hypothesize that RT is affected by the processes preceding the decision-making stage, e.g., encoding visual sensory information and extracting decision-relevant features from raw sensory information.
Keywords: behavioral response fluctuations; cortical network reorganization; functional brain network; perceptual decision-making task; reaction time.
Copyright © 2019 Maksimenko, Frolov, Hramov, Runnova, Grubov, Kurths and Pisarchik.
Figures




References
-
- Boccaletti S., Pisarchik A. N., del Genio C. I., Amann A. (2014). Synchronization: From Coupled Systems to Complex Networks. Cambridge, UK: Cambridge University Press.
LinkOut - more resources
Full Text Sources

