Absence of the Lectin Activation Pathway of Complement Ameliorates Proteinuria-Induced Renal Injury
- PMID: 31608060
- PMCID: PMC6768126
- DOI: 10.3389/fimmu.2019.02238
Absence of the Lectin Activation Pathway of Complement Ameliorates Proteinuria-Induced Renal Injury
Abstract
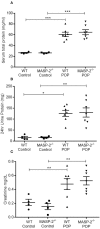
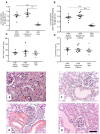
Proteinuria is an adverse prognostic feature in renal diseases. In proteinuric nephropathies, filtered proteins exert an injurious effect on the renal tubulointerstitium, resulting in inflammation and fibrosis. In the present study, we assessed to what extent complement activation via the lectin pathway may contribute to renal injury in response to proteinuria-related stress in proximal tubular cells. We used the well-established mouse model of protein overload proteinuria (POP) to assess the effect of lectin pathway inhibition on renal injury and fibrotic changes characteristic of proteinuric nephropathy. To this end, we compared experimental outcomes in wild type mice with MASP-2-deficient mice or wild type mice treated with MASP-2 inhibitor to block lectin pathway functional activity. Multiple markers of renal injury were assessed including renal function, proteinuria, macrophage infiltration, and cytokine release profiles. Both MASP-2-deficient and MASP-2 inhibitor-treated wild type mice exhibited renoprotection from proteinuria with significantly less tubulointerstitial injury when compared to isotype control antibody treated mice. This indicates that therapeutic targeting of MASP-2 in proteinuric nephropathies may offer a useful strategy in the clinical management of proteinuria associated pathologies in a variety of different underlying renal diseases.
Keywords: MASP; MBL-associated serine proteases; kidney; lectin pathway; nephropathy; proteinuria.
Copyright © 2019 Alghadban, Kenawy, Dudler, Schwaeble and Brunskill.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

