The interactions of dopamine and oxidative damage in the striatum of patients with neurodegenerative diseases
- PMID: 31613384
- PMCID: PMC6981021
- DOI: 10.1111/jnc.14898
The interactions of dopamine and oxidative damage in the striatum of patients with neurodegenerative diseases
Abstract
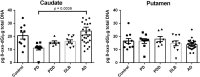
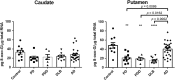
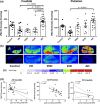
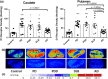
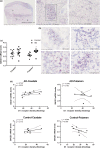
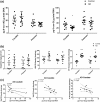
The striatum with a number of dopamine containing neurons, receiving projections from the substantia nigra and ventral tegmental area; plays a critical role in neurodegenerative diseases of motor and memory function. Additionally, oxidative damage to nucleic acid may be vital in the development of age-associated neurodegeneration. The metabolism of dopamine is recognized as one of the sources of reactive oxygen species through the Fenton mechanism. The proposed interactions of oxidative insults and dopamine in the striatum during the progression of diseases are the hypotheses of most interest to our study. This study investigated the possibility of significant interactions between these molecules that are involved in the late-stage of Alzheimer's disease (AD), Parkinson disease (PD), Parkinson disease dementia, dementia with Lewy bodies, and controls using ELISA assays, autoradiography, and mRNA in situ hybridization assay. Interestingly, lower DNA/RNA oxidative adducts levels in the caudate and putamen of diseased brains were observed with the exception of an increased DNA oxidative product in the caudate of AD brains. Similar changes were found for dopamine concentration and vesicular monoamine transporter 2 densities. We also found that downstream pre-synaptic dopamine D1 Receptor binding correlated with dopamine loss in Lewy body disease groups, and RNA damage and β-site APP cleaving enzyme 1 in the caudate of AD. This is the first demonstration of region-specific alterations of DNA/RNA oxidative damage which cannot be viewed in isolation, but rather in connection with the interrelationship between different neuronal events; chiefly DNA oxidative adducts and density of vesicular monoamine transporter 2 densities in AD and PD patients.
Keywords: Alzheimer's disease; Lewy body diseases; dopamine; oxidative damage; striatum.
© 2019 The Authors. Journal of Neurochemistry published by John Wiley & Sons Ltd on behalf of International Society for Neurochemistry.
Figures

Similar articles
-
Microglia Implicated in Tauopathy in the Striatum of Neurodegenerative Disease Patients from Genotype to Phenotype.Int J Mol Sci. 2020 Aug 22;21(17):6047. doi: 10.3390/ijms21176047. Int J Mol Sci. 2020. PMID: 32842621 Free PMC article.
-
Damage to dopamine systems differs between Parkinson's disease and Alzheimer's disease with parkinsonism.Ann Neurol. 1995 Mar;37(3):300-12. doi: 10.1002/ana.410370306. Ann Neurol. 1995. PMID: 7695230
-
Striatal dopaminergic markers in dementia with Lewy bodies, Alzheimer's and Parkinson's diseases: rostrocaudal distribution.Brain. 1999 Aug;122 ( Pt 8):1449-68. doi: 10.1093/brain/122.8.1449. Brain. 1999. PMID: 10430831
-
Biochemistry of postmortem brains in Parkinson's disease: historical overview and future prospects.J Neural Transm Suppl. 2007;(72):113-20. doi: 10.1007/978-3-211-73574-9_14. J Neural Transm Suppl. 2007. PMID: 17982884 Review.
-
Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse.Free Radic Biol Med. 2013 Sep;62:186-201. doi: 10.1016/j.freeradbiomed.2013.05.042. Epub 2013 Jun 3. Free Radic Biol Med. 2013. PMID: 23743292 Review.
Cited by
-
Dietary Curcumin Intake and Its Effects on the Transcriptome and Metabolome of Drosophila melanogaster.Int J Mol Sci. 2024 Jun 14;25(12):6559. doi: 10.3390/ijms25126559. Int J Mol Sci. 2024. PMID: 38928266 Free PMC article.
-
The Effect of Chronic Treatment with the Inhibitor of Phosphodiesterase 5 (PDE5), Sildenafil, in Combination with L-DOPA on Asymmetric Behavior and Monoamine Catabolism in the Striatum and Substantia Nigra of Unilaterally 6-OHDA-Lesioned Rats.Molecules. 2024 Sep 11;29(18):4318. doi: 10.3390/molecules29184318. Molecules. 2024. PMID: 39339313 Free PMC article.
-
Oxidative Stress and Neurodegeneration: Insights and Therapeutic Strategies for Parkinson's Disease.Neurol Int. 2024 Apr 29;16(3):502-517. doi: 10.3390/neurolint16030037. Neurol Int. 2024. PMID: 38804477 Free PMC article. Review.
-
Taurine protection attenuates bisphenol-A-induced behavioral, neurochemical, and histopathological alterations in male rats.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jul;398(7):8525-8542. doi: 10.1007/s00210-024-03767-4. Epub 2025 Jan 17. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39820545 Free PMC article.
-
Human striatal glia differentially contribute to AD- and PD-specific neurodegeneration.Nat Aging. 2023 Mar;3(3):346-365. doi: 10.1038/s43587-023-00363-8. Epub 2023 Feb 9. Nat Aging. 2023. PMID: 36993867 Free PMC article.
References
-
- Aarsland D. and Kurz M. W. (2010) The epidemiology of dementia associated with Parkinson disease. J. Neurol. Sci. 289, 18–22. - PubMed
-
- Abe T., Isobe C., Murata T., Sato C. and Tohgi H. (2003) Alteration of 8‐hydroxyguanosine concentrations in the cerebrospinal fluid and serum from patients with Parkinson's disease. Neurosci. Lett. 336, 105–108. - PubMed
-
- Bender A., Krishnan K. J., Morris C. M., et al (2006) High levels of mitochondrial DNA deletions in substantia nigra neurons in aging and Parkinson disease. Nat. Genet. 38, 515–517. - PubMed
-
- Bruns R. F., Mitchell S. N., Wafford K. A., et al (2018) Preclinical profile of a dopamine D1 potentiator suggests therapeutic utility in neurological and psychiatric disorders. Neuropharmacology 128, 351–365. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 AG066444/AG/NIA NIH HHS/United States
- R01 NS107281/NS/NINDS NIH HHS/United States
- S10 RR0227552/GF/NIH HHS/United States
- P01 AG026276/AG/NIA NIH HHS/United States
- R01 NS075321/NS/NINDS NIH HHS/United States
- R01 NS103957/NS/NINDS NIH HHS/United States
- R01 AG052550/AG/NIA NIH HHS/United States
- R01 NS058714/NS/NINDS NIH HHS/United States
- RF1 NS075321/NS/NINDS NIH HHS/United States
- R01 NS092865/NS/NINDS NIH HHS/United States
- P01 AG003991/AG/NIA NIH HHS/United States
- P50 AG005681/AG/NIA NIH HHS/United States
- R03 NS090214/NS/NINDS NIH HHS/United States
- R01 AG054567/AG/NIA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

