Interobserver, intraobserver, and interlaboratory variability in reporting pT4a colon cancer
- PMID: 31616981
- PMCID: PMC7028812
- DOI: 10.1007/s00428-019-02663-0
Interobserver, intraobserver, and interlaboratory variability in reporting pT4a colon cancer
Abstract
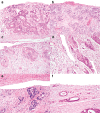
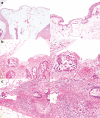
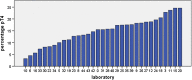
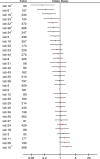
Clinical significance of the pT4 category in colon cancer is increasing with several therapeutic implications. The aim of this study was to evaluate variability in diagnosing pT4a colon cancer. Twelve pathologists classified 66 preselected scanned Hematoxylin/Eosin-stained slides with tumor cells at a distance of 25-1500 μm (n = 22), 0-25 μm (n = 22), or on (n = 22) the peritoneal surface. Inter- and intraobserver variability were calculated using Kappa statistics. For interlaboratory variability, pathology reports of pT3 and pT4a colon cancer were extracted from the Dutch Pathology Registry between 2012 and 2015. The proportion of pT4a (pT4a/(pT3+pT4a)) was compared between 33 laboratories. Potential risk of understaging was assessed by determining the average number of blocks taken from pT3 and pT4a N0-2M0 tumors with metachronous peritoneal metastasis. Interobserver variability among 12 pathologists was 0.50 (95%CI 0.41-0.60; moderate agreement). Intraobserver variability (8 pathologists) was 0.71 (substantial agreement). A total of 7745 reports with pT3 or pT4aN0-2M0 colon cancer from 33 laboratories were included for interlaboratory analysis. Median percentage of pT4a was 15.5% (range 3.2-24.6%). After adjustment for case mix, 8 labs diagnosed pT4a significantly less or more frequently than the median lab. Metachronous peritoneal metastases were histologically verified in 170 of 6629 pT3 and in 129 of 1116 pT4a tumors, with a mean number of blocks of 4.03(SD 1.51) and 4.78 (SD 1.76) taken from the primary tumors, respectively (p < 0.001). A substantial variability in diagnosing pT4a colon cancer exists, both at pathologist and laboratory level. Diagnosis of pT4a stage appears to be challenging and there is a need for standardizing assessment of this pathological entity.
Keywords: Interobserver variability; T4 colon cancer, peritoneal tumor involvement.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



References
-
- Klaver CEL, Gietelink L, Bemelman WA, Wouters M, Tollenaar RAEM, Tanis PJ. Locally advanced colon cancer; current clinical practice and treatment outcome in the Netherlands. Color Dis. 2015;17:23. - PubMed
-
- Brierly J, Gospodarowicz M, Wittekind C. The TNM classification of malignant tumours. 8. Oxford: Wiley Blackwell; 2017.
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

