Ni-Catalyzed Suzuki-Miyaura Cross-Coupling of Aliphatic Amides on the Benchtop
- PMID: 31621338
- PMCID: PMC6994262
- DOI: 10.1021/acs.orglett.9b03434
Ni-Catalyzed Suzuki-Miyaura Cross-Coupling of Aliphatic Amides on the Benchtop
Abstract
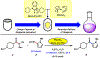
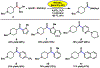
Suzuki-Miyaura cross-couplings of amides offer an approach to the synthesis of ketones that avoids the use of basic or pyrophoric nucleophiles. However, these reactions require glovebox manipulations, thus limiting their practicality. We report a benchtop protocol for Suzuki-Miyaura cross-couplings of aliphatic amides that utilizes a paraffin capsule containing a Ni(0) precatalyst and NHC ligand. This methodology is broad in scope, is scalable, and provides a user-friendly approach to convert aliphatic amides to alkyl-aryl ketones.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Nickel-catalysed Suzuki-Miyaura coupling of amides.Nat Chem. 2016 Jan;8(1):75-9. doi: 10.1038/nchem.2388. Epub 2015 Nov 9. Nat Chem. 2016. PMID: 26673267
-
Reductive Arylation of Amides via a Nickel-Catalyzed Suzuki-Miyaura-Coupling and Transfer-Hydrogenation Cascade.Angew Chem Int Ed Engl. 2021 Feb 1;60(5):2472-2477. doi: 10.1002/anie.202012048. Epub 2020 Nov 30. Angew Chem Int Ed Engl. 2021. PMID: 33029868 Free PMC article.
-
Nickel-Catalyzed Suzuki-Miyaura Coupling of Aliphatic Amides.ACS Catal. 2018 Feb 2;8(2):1003-1008. doi: 10.1021/acscatal.7b03688. Epub 2017 Dec 20. ACS Catal. 2018. PMID: 29682398 Free PMC article.
-
Suzuki-miyaura cross-coupling in acylation reactions, scope and recent developments.Molecules. 2013 Jan 17;18(1):1188-213. doi: 10.3390/molecules18011188. Molecules. 2013. PMID: 23344208 Free PMC article. Review.
-
Temperature-Controlled Mechanochemistry Unlocks the Nickel-Catalyzed Suzuki-Miyaura-Type Coupling of Aryl Sulfamates at Different Scales.Angew Chem Int Ed Engl. 2023 Jan 9;62(2):e202215094. doi: 10.1002/anie.202215094. Epub 2022 Dec 2. Angew Chem Int Ed Engl. 2023. PMID: 36331906 Review.
Cited by
-
Amide N-C Bond Activation: A Graphical Overview of Acyl and Decarbonylative Coupling.SynOpen. 2023 Feb;7(1):88-101. doi: 10.1055/a-2035-6733. Epub 2023 Mar 6. SynOpen. 2023. PMID: 38037650 Free PMC article.
-
Structurally Diverse Bench-Stable Nickel(0) Pre-Catalysts: A Practical Toolkit for In Situ Ligation Protocols.Angew Chem Int Ed Engl. 2023 Feb 20;62(9):e202211794. doi: 10.1002/anie.202211794. Epub 2023 Jan 18. Angew Chem Int Ed Engl. 2023. PMID: 36524997 Free PMC article.
-
Easily Accessible and Solution-Stable Ni(0) Precatalysts for High-Throughput Experimentation.Chemistry. 2025 Feb 25;31(12):e202403960. doi: 10.1002/chem.202403960. Epub 2025 Jan 28. Chemistry. 2025. PMID: 39822108 Free PMC article.
-
Activation of C-O and C-N Bonds Using Non-Precious-Metal Catalysis.ACS Catal. 2020 Oct 16;10(20):12109-12126. doi: 10.1021/acscatal.0c03334. Epub 2020 Sep 10. ACS Catal. 2020. PMID: 33868770 Free PMC article. No abstract available.
-
Enantioselective nickel-catalyzed Mizoroki-Heck cyclizations of amide electrophiles.Chem Sci. 2024 Jan 8;15(7):2593-2600. doi: 10.1039/d3sc05797f. eCollection 2024 Feb 14. Chem Sci. 2024. PMID: 38362425 Free PMC article.
References
-
- Larock RC Comprehensive Organic Transformations: A Guide to Functional Group Preparations; VCH: New York, 1989; pp 685–702.
- Nicholson JW; Wilson AD The conversion of carboxylic acids to ketones: a repeated discovery. J. Chem. Educ 2004, 81, 1362–1366.
- Amani J; Molander GA Direct conversion of carboxylic acids to alkyl ketones. Org. Lett 2017, 19, 3612–3615. - PMC - PubMed
-
- Nahm S; Weinreb SM N-Methoxy-N-methylamides as effective acylating agents. Tetrahedron Lett 1981, 22, 3815–3818.
- Balasubramaniam S; Aidhen IS The growing synthetic utility of the Weinreb amide. Synthesis 2008, 3707–3738.
-
- For couplings of acyl halides, anhydrides, esters, and thioesters, see:
- Labadie JW; Tueting D; Stille JK Synthetic utility of the palladium-catalyzed coupling reaction of acid chlorides with organotins. J. Org. Chem 1983, 48, 4634–4642.
- Stille JK The palladium-catalyzed cross-coupling reactions of organotin reagents with organic electrophiles. Angew. Chem., Int. Ed 1986, 25, 508–524.
- Stephan MS; Teunissen AJJM; de Vries JG Heck reactions without salt formation: aromatic carboxylic anhydrides as arylating agents. Angew. Chem., Int. Ed 1998, 37, 662–664. - PubMed
- Gooßen LJ; Ghosh K Palladium-catalyzed synthesis of aryl ketones from boronic acids and carboxylic acids or anhydrides. Angew. Chem., Int. Ed 2001, 40, 3458–3460. - PubMed
- Kakino R; Narahashi H; Shimizu I; Yamamoto A Palladium-catalyzed direct conversion of carboxylic acids into ketones with organoboronic acids promoted by anhydride activators. Bull. Chem. Soc. Jpn 2002, 75, 1333–1345.
- Bykov VV; Korolev DN; Bumagin NA Palladium-catalyzed reactions of organoboron compounds with acyl chlorides. Russ. Chem. Bull 1997, 46, 1631–1632.
- Zhang L; Wu J; Shi L; Xia C; Li F Ionically tagged benzimidazole palladium(II) complex: preparation and catalytic application in cross-coupling reactions. Tetrahedron Lett 2011, 52, 3897–3901.
- Kakino R; Shimizu I; Yamamoto A Synthesis of trifluoromethyl ketones by palladium-catalyzed cross-coupling reaction of phenyl trifluoroacetate with organoboron compounds. Bull. Chem. Soc. Jpn 2001, 74, 371–376.
- Cherney AH; Reisman SE Pd-catalyzed Fukuyama cross-coupling of secondary organozinc reagents for the direct synthesis of unsymmetrical ketones. Tetrahedron 2014, 70, 3259–3265.
- Harada T; Kotani Y; Katsuhira T; Oku A Novel method for generation of secondary organozinc reagent: application to tandem carbon-carbon bond formation reaction of 1,1-dibromoalkane. Tetrahedron Lett 1991, 32, 1573–1576.
- Negishi E-I; Bagheri V; Chatterjee S; Luo F-T; Miller JA; Stoll AT Palladium-catalyzed acylation of organozincs and other organometallics as a convenient route to ketones. Tetrahedron Lett 1983, 24, 5181–5184.
- Iwai T; Nakai T; Mihara M; Ito T; Mizuno T; Ohno T Pd-catalyzed cross-coupling reactions of pyridine carboxylic acid chlorides with alkylzinc reagents. Synlett 2009, 1091–1094.
- Grey RA A palladium-catalyzed synthesis of ketones from acid chlorides and organozinc compounds. J. Org. Chem 1984, 49, 2288–2289.
- Bercot EA; Rovis T A mild and efficient catalytic alkylative monofunctionalization of cyclic anhydrides. J. Am. Chem. Soc 2002, 124, 174–175. - PubMed
- Bercot EA; Rovis T A palladium-catalyzed enantioselective alkylative desymmetrization of meso-succinic anhydrides. J. Am. Chem. Soc 2004, 126, 10248–10249. - PubMed
- Johnson JB; Yu RT; Fink P; Bercot EA; Rovis T Selective substituent transfer from mixed zinc reagents in Ni-catalyzed anhydride alkylation. Org. Lett 2006, 8, 4307–4310. - PubMed
- Tokuyama H; Yokoshima S; Yamashita T; Fukuyama T A novel ketone synthesis by a palladium-catalyzed reaction of thiol esters and organozinc reagents. Tetrahedron Lett 1998, 39, 3189–3192.
- Mori Y; Seki M A novel procedure for the synthesis of multifunctional ketones through the Fukuyama coupling reaction employing dialkylzincs. Tetrahedron Lett 2004, 45, 7343–7345.
- Miyazaki T; Han-ya Y; Tokuyama H; Fukuyama T New odorless protocols for the synthesis of aldehydes and ketones from thiol esters. Synlett 2004, 3, 477–480.
- Dieter RK Reaction of acyl chlorides with organometallic reagents: a banquet table of metals for ketone synthesis. Tetrahedron 1999, 55, 4177–4236.
- Shi S; Nolan SP; Szostak M Well-defined palladium(II)-NHC precatalysts for cross-coupling reactions of amides and esters by selective N–C/O–C cleavage. Acc. Chem. Res 2018, 51, 2589–2599. - PubMed
- Ben Halima T; Zhang W; Yalaoui T; Hong X; Yang Y-F; Houk KN; Newman SG Palladium-catalyzed Suzuki–Miyaura coupling of aryl esters. J. Am. Chem. Soc 2017, 139, 1311–1318. - PubMed
-
- For recent reviews, see:
- Dander JE; Garg NK Breaking amides using nickel catalysis. ACS Catal 2017, 7, 1413–1423. - PMC - PubMed
- Meng G; Shi S; Szostak M Cross-coupling of amides by N–C bond activation. Synlett 2016, 2530–2540.
- Takise R; Muto K; Yamaguchi J Cross-coupling of aromatic esters and amides. Chem. Soc. Rev 2017, 46, 5864–5888. - PubMed
- Kaiser D; Bauer A; Lemmerer M; Maulide N Chem. Soc. Rev 2018, 47, 7899–7925. - PubMed
-
- For nickel-catalyzed reactions proceeding with cleavage of the amide C–N bond, see:
- Hie L; Fine Nathel NF; Shah TK; Baker EL; Hong X; Yang Y-F; Liu P; Houk KN; Garg NK Conversion of amides to esters by the nickel-catalysed activation of amide C-N bonds. Nature 2015, 524, 79–83. - PMC - PubMed
- Weires NA; Baker EL; Garg NK Nickel-catalysed Suzuki–Miyaura coupling of amides. Nat. Chem 2016, 8, 75–79. - PubMed
- Baker EL; Yamano MM; Zhou Y; Anthony SM; Garg NK A two-step approach to achieve secondary amide transamidation enabled by nickel catalysis. Nat. Commun 2016, 7, 11554. - PMC - PubMed
- Simmons BJ; Weires NA; Dander JE; Garg NK Nickel-catalyzed alkylation of amide derivatives. ACS Catal 2016, 6, 3176–3179. - PMC - PubMed
- Shi S; Szostak M Nickel-catalyzed diaryl ketone synthesis by N–C bond cleavage: direct Negishi cross-coupling of primary amides by site-selective N,N-di-Boc activation. Org. Lett 2016, 18, 5872–5875. - PubMed
- Shi S; Szostak M Efficient synthesis of diaryl ketones by nickel-catalyzed Negishi cross-coupling of amides via carbon–nitrogen bond cleavage at room temperature accelerated by solvent effect. Chem. Eur. J 2016, 22, 10420–10424. - PubMed
- Hie L; Baker EL; Anthony SM; Desrosiers J-N; Senanayake C; Garg NK Nickel-catalyzed esterification of aliphatic amides. Angew. Chem., Int. Ed 2016, 55, 15129–15132. - PMC - PubMed
- Dey A; Sasmal S; Seth K; Lahiri GK; Maiti D Nickel-catalyzed deamidative step-down reduction of amides to aromatic hydrocarbons. ACS Catal 2017, 7, 433–437.
- Ni S; Zhang W; Mei H; Han J; Pan Y Ni-catalyzed reductive cross-coupling of amides with aryl iodide electrophiles via C–N bond activation. Org. Lett 2017, 19, 2536–2539. - PubMed
- Medina JM; Moreno J; Racine S; Du S; Garg NK Mizoroki–Heck cyclizations of amide derivatives for the introduction of quaternary centers. Angew. Chem., Int. Ed 2017, 56, 6567–6571. - PMC - PubMed
- Hu J; Wang M; Pu X; Shi Z Nickel-catalysed retro-hydroamidocarbonylation of aliphatic amides to olefins. Nat. Commun 2017, 8, 14993. - PMC - PubMed
- Weires NA; Caspi DD; Garg NK Kinetic modeling of the nickel-catalyzed esterification of amides. ACS Catal 2017, 7, 4381–4385. - PMC - PubMed
- Shi S; Szostak M Nickel-catalyzed Negishi cross-coupling of N-acylsuccinimides: stable, amide-based, twist-controlled acyl transfer reagents via N–C activation. Synthesis 2017, 3602–3608.
- Dander JE; Baker EL; Garg NK Nickel-catalyzed transamidation of aliphatic amide derivatives. Chem. Sci 2017, 8, 6433–6438. - PMC - PubMed
- Huang P-Q; Chen H Ni-catalyzed cross-coupling reactions of N-acylpyrrole-type amides with organoboron reagents. Chem. Commun 2017, 53, 12584–12587. - PubMed
- Deguchi T; Xin H-L; Morimoto H; Ohshima T Direct catalytic alcoholysis of unactivated 8-aminoquinoline amides. ACS Catal 2017, 7, 3157–3161.
- Dander JE; Giroud M; Racine S; Darzi ER; Alvizo O; Entwistle D; Garg NK Chemoenzymatic conversion of amides to enantioenriched alcohols in aqueous medium. Commun. Chem 2019, 2, 82. - PMC - PubMed

