Protein Kinase C and Calmodulin Serve As Calcium Sensors for Calcium-Stimulated Endocytosis at Synapses
- PMID: 31628181
- PMCID: PMC6880464
- DOI: 10.1523/JNEUROSCI.0182-19.2019
Protein Kinase C and Calmodulin Serve As Calcium Sensors for Calcium-Stimulated Endocytosis at Synapses
Abstract
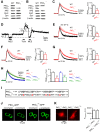
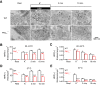
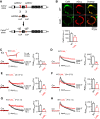
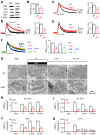
Calcium influx triggers and facilitates endocytosis, which recycles vesicles and thus sustains synaptic transmission. Despite decades of studies, the underlying calcium sensor remained not well understood. Here, we examined two calcium binding proteins, protein kinase C (PKC) and calmodulin. Whether PKC is involved in endocytosis was unclear; whether calmodulin acts as a calcium sensor for endocytosis was neither clear, although calmodulin involvement in endocytosis had been suggested. We generated PKC (α or β-isoform) and calmodulin (calmodulin 2 gene) knock-out mice of either sex and measured endocytosis with capacitance measurements, pHluorin imaging and electron microscopy. We found that these knock-outs inhibited slow (∼10-30 s) and rapid (<∼3 s) endocytosis at large calyx-type calyces, and inhibited slow endocytosis and bulk endocytosis (forming large endosome-like structures) at small conventional hippocampal synapses, suggesting the involvement of PKC and calmodulin in three most common forms of endocytosis-the slow, rapid and bulk endocytosis. Inhibition of slow endocytosis in PKC or calmodulin 2 knock-out hippocampal synapses was rescued by overexpressing wild-type PKC or calmodulin, but not calcium-binding-deficient PKC or calmodulin mutant, respectively, suggesting that calcium stimulates endocytosis by binding with its calcium sensor PKC and calmodulin. PKC and calmodulin 2 knock-out inhibited calcium-dependent vesicle mobilization to the readily releasable pool, suggesting that PKC and calmodulin may mediate calcium-dependent facilitation of vesicle mobilization. These findings shed light on the molecular signaling link among calcium, endocytosis and vesicle mobilization that are crucial in maintaining synaptic transmission and neuronal network activity.SIGNIFICANCE STATEMENT Vesicle fusion releases neurotransmitters to mediate synaptic transmission. To sustain synaptic transmission, fused vesicles must be retrieved via endocytosis. Accumulating evidence suggests that calcium influx triggers synaptic vesicle endocytosis. However, how calcium triggers endocytosis is not well understood. Using genetic tools together with capacitance measurements, optical imaging and electron microscopy, we identified two calcium sensors, including protein kinase C (α and β isoforms) and calmodulin, for the most commonly observed forms of endocytosis: slow, rapid, and bulk. We also found that these two proteins are involved in calcium-dependent vesicle mobilization to the readily releasable pool. These results provide the molecular signaling link among calcium, endocytosis, and vesicle mobilization that are essential in sustaining synaptic transmission and neuronal network activity.
Keywords: calmodulin; capacitance measurement; electron microscopy; endocytosis; pHluorin imaging; protein kinase C.
Copyright © 2019 the authors.
Figures



Similar articles
-
Ca(2+) and calmodulin initiate all forms of endocytosis during depolarization at a nerve terminal.Nat Neurosci. 2009 Aug;12(8):1003-1010. doi: 10.1038/nn.2355. Epub 2009 Jul 26. Nat Neurosci. 2009. PMID: 19633667 Free PMC article.
-
Myosin light chain kinase accelerates vesicle endocytosis at the calyx of Held synapse.J Neurosci. 2014 Jan 1;34(1):295-304. doi: 10.1523/JNEUROSCI.3744-13.2014. J Neurosci. 2014. PMID: 24381290 Free PMC article.
-
Actin Is Crucial for All Kinetically Distinguishable Forms of Endocytosis at Synapses.Neuron. 2016 Dec 7;92(5):1020-1035. doi: 10.1016/j.neuron.2016.10.014. Epub 2016 Nov 10. Neuron. 2016. PMID: 27840001 Free PMC article.
-
Ca2+ Dependence of Synaptic Vesicle Endocytosis.Neuroscientist. 2016 Oct;22(5):464-76. doi: 10.1177/1073858415588265. Epub 2015 May 21. Neuroscientist. 2016. PMID: 25998187 Review.
-
Ca2+-dependent regulation of synaptic vesicle endocytosis.Neurosci Res. 2012 May;73(1):1-7. doi: 10.1016/j.neures.2012.02.012. Epub 2012 Mar 3. Neurosci Res. 2012. PMID: 22401840 Review.
Cited by
-
A positive feedback loop between Flower and PI(4,5)P2 at periactive zones controls bulk endocytosis in Drosophila.Elife. 2020 Dec 10;9:e60125. doi: 10.7554/eLife.60125. Elife. 2020. PMID: 33300871 Free PMC article.
-
Multiple Modes of Fusion and Retrieval at the Calyx of Held Synapse.Adv Neurobiol. 2023;33:43-62. doi: 10.1007/978-3-031-34229-5_3. Adv Neurobiol. 2023. PMID: 37615863
-
The role of CaMKK2 in Golgi-associated vesicle trafficking.Biochem Soc Trans. 2023 Feb 27;51(1):331-342. doi: 10.1042/BST20220833. Biochem Soc Trans. 2023. PMID: 36815702 Free PMC article. Review.
-
Calmodulin Triggers Activity-Dependent rRNA Biogenesis via Interaction with DDX21.J Neurosci. 2024 Aug 28;44(35):e1841232024. doi: 10.1523/JNEUROSCI.1841-23.2024. J Neurosci. 2024. PMID: 39060175 Free PMC article.
-
Dynamin 1 controls vesicle size and endocytosis at hippocampal synapses.Cell Calcium. 2022 May;103:102564. doi: 10.1016/j.ceca.2022.102564. Epub 2022 Feb 18. Cell Calcium. 2022. PMID: 35220002 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
