CuII (atsm) inhibits ferroptosis: Implications for treatment of neurodegenerative disease
- PMID: 31655003
- PMCID: PMC7012947
- DOI: 10.1111/bph.14881
CuII (atsm) inhibits ferroptosis: Implications for treatment of neurodegenerative disease
Abstract
Background and purpose: Diacetyl-bis(4-methyl-3-thiosemicarbazonato)copperII (CuII (atsm)) ameliorates neurodegeneration and delays disease progression in mouse models of amyotrophic lateral sclerosis (ALS) and Parkinson's disease (PD), yet the mechanism of action remains uncertain. Promising results were recently reported for separate Phase 1 studies in ALS patients and PD patients. Affected tissue in these disorders shares features of elevated Fe, low glutathione and increased lipid peroxidation consistent with ferroptosis, a novel form of regulated cell death. We therefore evaluated the ability of CuII (atsm) to inhibit ferroptosis.
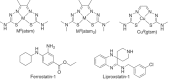
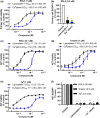
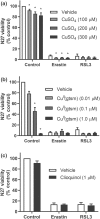
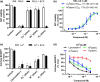
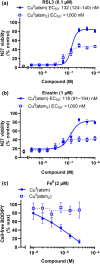
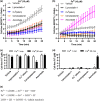
Experimental approach: Ferroptosis was induced in neuronal cell models by inhibition of glutathione peroxidase-4 activity with RSL3 or by blocking cystine uptake with erastin. Cell viability and lipid peroxidation were assessed and the efficacy of CuII (atsm) was compared to the known antiferroptotic compound liproxstatin-1.
Key results: CuII (atsm) protected against lipid peroxidation and ferroptotic lethality in primary and immortalised neuronal cell models (EC50 : ≈130 nM, within an order of magnitude of liproxstatin-1). NiII (atsm) also prevented ferroptosis with similar potency, whereas ionic CuII did not. In cell-free systems, CuII (atsm) and NiII (atsm) inhibited FeII -induced lipid peroxidation, consistent with these compounds quenching lipid radicals.
Conclusions and implications: The antiferroptotic activity of CuII (atsm) could therefore be the disease-modifying mechanism being tested in ALS and PD trials. With potency in vitro approaching that of liproxstatin-1, CuII (atsm) possesses favourable properties such as oral bioavailability and entry into the brain that make it an attractive investigational product for clinical trials of ferroptosis-related diseases.
© 2019 The British Pharmacological Society.
Conflict of interest statement
A.I.B. is a shareholder in Prana Biotechnology Ltd, Cogstate Ltd, Brighton Biotech LLC, Grunbiotics Pty Ltd, Eucalyptus Pty Ltd, and Mesoblast Ltd. He is a paid consultant for, and has a profit share interest in, Collaborative Medicinal Development Pty Ltd. P.S.D. has served as a consultant to Collaborative Medicinal Development LLC. Collaborative Medicinal Development LLC has licensed intellectual property related to this subject from The University of Melbourne, where the inventors include P.S.D. and K.J.B.
Figures
References
-
- Adlard, P. A. , Cherny, R. A. , Finkelstein, D. I. , Gautier, E. , Robb, E. , Cortes, M. , … Bush, A. I. (2008). Rapid restoration of cognition in Alzheimer's transgenic mice with 8‐hydroxy quinoline analogs is associated with decreased interstitial Aβ. Neuron, 59, 43–55. 10.1016/j.neuron.2008.06.018 - DOI - PubMed
-
- Ayton, S. , Lei, P. , Hare, D. J. , Duce, J. A. , George, J. L. , Adlard, P. A. , … Bush, A. I. (2015). Parkinson's disease iron deposition caused by nitric oxide‐induced loss of β‐amyloid precursor protein. The Journal of Neuroscience, 35, 3591–3597. 10.1523/JNEUROSCI.3439-14.2015 - DOI - PMC - PubMed
-
- Beraldo, H. , Boyd, L. P. , & West, D. X. (1998). Copper(II) and nickel(II) complexes of glyoxaldehyde bis(N(3)‐substituted thiosemicarbazones). Transition Metal Chemistry, 23, 67–71.
-
- Cherubini, A. , Ruggiero, C. , Polidori, M. C. , & Mecocci, P. (2005). Potential markers of oxidative stress in stroke. Free Radical Biology & Medicine, 39, 841–852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

