Population pharmacokinetics and pharmacodynamics of dobutamine in neonates on the first days of life
- PMID: 31657867
- PMCID: PMC7015735
- DOI: 10.1111/bcp.14146
Population pharmacokinetics and pharmacodynamics of dobutamine in neonates on the first days of life
Abstract
Aims: To describe the pharmacokinetics (PK) and concentration-related effects of dobutamine in critically ill neonates in the first days of life, using nonlinear mixed effects modelling.
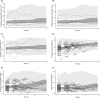
Methods: Dosing, plasma concentration and haemodynamic monitoring data from a dose-escalation study were analysed with a simultaneous population PK and pharmacodynamic model. Neonates receiving continuous infusion of dobutamine 5-20 μg kg-1 min-1 were included. Left ventricular ejection fraction (LVEF) and cardiac output of right and left ventricle (RVO, LVO) were measured on echocardiography; heart rate (HR), mean arterial pressure (MAP), peripheral arterial oxygen saturation and cerebral regional oxygen saturation were recorded from patient monitors.
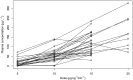
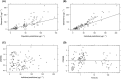
Results: Twenty-eight neonates with median (range) gestational age of 30.4 (22.7-41.0) weeks and birth weight (BW) of 1618 (465-4380) g were included. PK data were adequately described by 1-compartmental linear structural model. Dobutamine clearance (CL) was described by allometric scaling on BW with sigmoidal maturation function of postmenstrual age (PMA). The final population PK model parameter mean typical value (standard error) estimates, standardised to median BW of 1618 g, were 41.2 (44.5) L h-1 for CL and 5.29 (0.821) L for volume of distribution, which shared a common between subject variability of 29% (17.2%). The relationship between dobutamine concentration and RVO/LVEF was described by linear model, between concentration and LVO/HR/MAP/cerebral fractional tissue oxygen extraction by sigmoidal Emax model.
Conclusion: In the postnatal transitional period, PK of dobutamine was described by a 1-compartmental linear model, CL related to BW and PMA. A concentration-response relationship with haemodynamic variables has been established.
Keywords: cardiovascular pharmacology, intensive care, neonatology, NONMEM, pharmacokinetic-pharmacodynamic.
© 2019 The Authors. British Journal of Clinical Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Figures

References
-
- Tuttle RR, Mills J. Dobutamine: development of a new catecholamine to selectively increase cardiac contractility. Circ Res. 1975;36(1):185‐196. - PubMed
-
- Burns ML, Stensvold HJ, Risnes K, et al. Inotropic therapy in newborns, a population‐based national registry study. Pediatr Crit Care Med. 2016;17(10):948‐956. - PubMed
-
- Osborn DA, Evans N, Kluckow M. Left ventricular contractility in extremely premature infants in the first day and response to inotropes. Pediatr Res. 2007;61(3):335‐340. - PubMed
-
- Brew N, Nakamura S, Hale N, et al. Dobutamine treatment reduces inflammation in the preterm fetal sheep brain exposed to acute hypoxia. Pediatr Res. 2018;84(3):442‐450. - PubMed
-
- Durrmeyer X, Marchand‐Martin L, Porcher R, et al. Abstention or intervention for isolated hypotension in the first 3 days of life in extremely preterm infants: association with short‐term outcomes in the EPIPAGE 2 cohort study. Arch Dis Child ‐ Fetal Neonatal Ed. 2017;102(6):490‐496. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

