Population pharmacokinetics of a triple-secured fibrinogen concentrate administered to afibrinogenaemic patients: Observed age- and body weight-related differences and consequences for dose adjustment in children
- PMID: 31658379
- PMCID: PMC7015754
- DOI: 10.1111/bcp.14147
Population pharmacokinetics of a triple-secured fibrinogen concentrate administered to afibrinogenaemic patients: Observed age- and body weight-related differences and consequences for dose adjustment in children
Abstract
Aims: The pharmacokinetics (PK) of a triple-secured fibrinogen concentrate (FC) was assessed in patients ≥40 kg by noncompartmental analysis over a period of 14 days with multiple blood samples. Limited PK time point assessments in children lead to consideration of using Bayesian estimation for paediatric data. The objectives were (i) to define the population PK of FC in patients with afibrinogenaemia; (ii) to detect age- and body weight-related differences and consequences for dose adjustment.
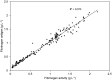
Methods: A population PK model was built using plasma fibrinogen activity data collected in 31 patients aged 1 to 48 years who had participated in a single-dose PK study with FC 0.06 g kg-1 .
Results: A 1-compartment model with allometric scaling accounting for body weight was found to best describe the kinetics of FC. Addition of age and sex as covariates did not improve the model. Incremental in vivo recovery assessed at the end of infusion with the predicted maximal concentrations was lower, weight-adjusted clearance was higher, and fibrinogen elimination half-life was shorter in patients <40 kg than patients ≥40 kg. Interpatient variability was similar in both groups.
Conclusion: Dosing in patients ≥40 kg based on the previous empirical finding using noncompartmental analysis where FC 1 g kg-1 raises the plasma fibrinogen activity by 23 g L-1 was confirmed. In patients <40 kg, (covering the age range from birth up to about 12 years old) FC 1 g kg-1 raises the plasma fibrinogen by 19 g L-1 . Dosing should be adapted accordingly unless therapy is individualized.
Keywords: coagulation, congenital disorders, NONMEM, paediatrics, population analysis.
© 2019 LFB Biotechnologies. British Journal of Clinical Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Conflict of interest statement
The study was funded by LFB, les Ulis, France. A.B., O.R., J.L., W.S., A.D., M.B.T., D.G., C.H. and F.B. were full‐time employees of LFB. E.F. received financial compensation for their involvement in the study.
Figures



References
-
- Lak M, Keihani M, Elahi F, Peyvandi F, Mannucci PM. Bleeding and thrombosis in 55 patients with inherited afibrinigenaemia. Br J Haematol. 1999;107(1):204‐206. - PubMed
-
- Mannucci PM, Duga S, Peyvandi F. Recessively inherited coagulation disorders. Blood. 2004;104(5):1243‐1252. - PubMed
-
- Samama M, Conard J, Horellou MH, Lecompte T. Physiologie et exploration de l'hémostase. Doin éditeurs. 1990;153‐155.
-
- de Moerloose P, Neerman‐Arbez M. Treatment of congenital fibrinogen disorders. Expert Opin Biol Ther. 2008;8(7):979‐992. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

