Hair cell maturation is differentially regulated along the tonotopic axis of the mammalian cochlea
- PMID: 31661723
- PMCID: PMC6972525
- DOI: 10.1113/JP279012
Hair cell maturation is differentially regulated along the tonotopic axis of the mammalian cochlea
Abstract
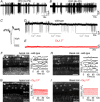
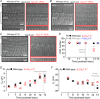
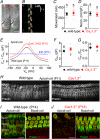
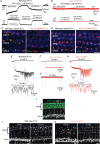
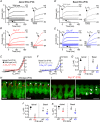
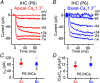
Key points: Outer hair cells (OHCs) enhance the sensitivity and the frequency tuning of the mammalian cochlea. Similar to the primary sensory receptor, the inner hair cells (IHCs), the mature functional characteristics of OHCs are acquired before hearing onset. We found that OHCs, like IHCs, fire spontaneous Ca2+ -induced action potentials (APs) during immature stages of development, which are driven by CaV 1.3 Ca2+ channels. We also showed that the development of low- and high-frequency hair cells is differentially regulated during pre-hearing stages, with the former cells being more strongly dependent on experience-independent Ca2+ action potential activity.
Abstract: Sound amplification within the mammalian cochlea depends upon specialized hair cells, the outer hair cells (OHCs), which possess both sensory and motile capabilities. In various altricial rodents, OHCs become functionally competent from around postnatal day 7 (P7), before the primary sensory inner hair cells (IHCs), which become competent at about the onset of hearing (P12). The mechanisms responsible for the maturation of OHCs and their synaptic specialization remain poorly understood. We report that spontaneous Ca2+ activity in the immature cochlea, which is generated by CaV 1.3 Ca2+ channels, differentially regulates the maturation of hair cells along the cochlea. Under near-physiological recording conditions we found that, similar to IHCs, immature OHCs elicited spontaneous Ca2+ action potentials (APs), but only during the first few postnatal days. Genetic ablation of these APs in vivo, using CaV 1.3-/- mice, prevented the normal developmental acquisition of mature-like basolateral membrane currents in low-frequency (apical) hair cells, such as IK,n (carried by KCNQ4 channels), ISK2 and IACh (α9α10nAChRs) in OHCs and IK,n and IK,f (BK channels) in IHCs. Electromotility and prestin expression in OHCs were normal in CaV 1.3-/- mice. The maturation of high-frequency (basal) hair cells was also affected in CaV 1.3-/- mice, but to a much lesser extent than apical cells. However, a characteristic feature in CaV 1.3-/- mice was the reduced hair cell size irrespective of their cochlear location. We conclude that the development of low- and high-frequency hair cells is differentially regulated during development, with apical cells being more strongly dependent on experience-independent Ca2+ APs.
Keywords: action potentials; auditory; calcium signals; cochlea; development; hair cells.
© 2019 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures




Comment in
-
A maturing view of cochlear calcium.J Physiol. 2020 Jan;598(1):7-8. doi: 10.1113/JP279215. Epub 2019 Dec 26. J Physiol. 2020. PMID: 31745992 No abstract available.
References
-
- Ashmore J (2008). Cochlear outer hair cell motility. Physiol Rev 88, 173–210. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

