Transplantation of Mouse Induced Pluripotent Stem Cell-Derived Podocytes in a Mouse Model of Membranous Nephropathy Attenuates Proteinuria
- PMID: 31664077
- PMCID: PMC6820764
- DOI: 10.1038/s41598-019-51770-0
Transplantation of Mouse Induced Pluripotent Stem Cell-Derived Podocytes in a Mouse Model of Membranous Nephropathy Attenuates Proteinuria
Retraction in
-
Retraction Note: Transplantation of Mouse Induced Pluripotent Stem Cell-Derived Podocytes in a Mouse Model of Membranous Nephropathy Attenuates Proteinuria.Sci Rep. 2021 Jul 5;11(1):13831. doi: 10.1038/s41598-021-93174-z. Sci Rep. 2021. PMID: 34226599 Free PMC article. No abstract available.
Abstract
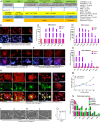
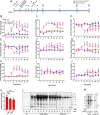
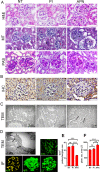
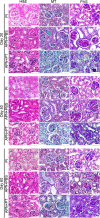
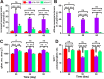
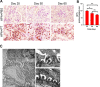
Injury to podocytes is a principle cause of initiation and progression of both immune and non-immune mediated glomerular diseases that result in proteinuria and decreased function of the kidney. Current advances in regenerative medicine shed light on the therapeutic potential of cell-based strategies for treatment of such disorders. Thus, there is hope that generation and transplantation of podocytes from induced pluripotent stem cells (iPSCs), could potentially be used as a curative treatment for glomerulonephritis caused by podocytes injury and loss. Despite several reports on the generation of iPSC-derived podocytes, there are rare reports about successful use of these cells in animal models. In this study, we first generated a model of anti-podocyte antibody-induced heavy proteinuria that resembled human membranous nephropathy and was characterized by the presence of sub-epithelial immune deposits and podocytes loss. Thereafter, we showed that transplantation of functional iPSC-derived podocytes following podocytes depletion results in recruitment of iPSC-derived podocytes within the damaged glomerulus, and leads to attenuation of proteinuria and histological alterations. These results provided evidence that application of iPSCs-derived renal cells could be a possible therapeutic strategy to favorably influence glomerular diseases outcomes.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Meliambro, K., He, J. C. & Campbell, K. N. The podocyte as a therapeutic target in proteinuric kidney disease. Journal of Nephrology & Therapeutics2013 (2013).
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

