Neurovascular Coupling under Chronic Stress Is Modified by Altered GABAergic Interneuron Activity
- PMID: 31672788
- PMCID: PMC6978951
- DOI: 10.1523/JNEUROSCI.1357-19.2019
Neurovascular Coupling under Chronic Stress Is Modified by Altered GABAergic Interneuron Activity
Abstract
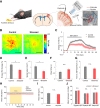
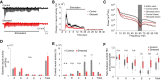
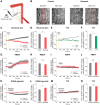
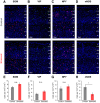
Neurovascular coupling (NVC), the interaction between neural activity and vascular response, ensures normal brain function by maintaining brain homeostasis. We previously reported altered cerebrovascular responses during functional hyperemia in chronically stressed animals. However, the underlying neuronal-level changes associated with those hemodynamic changes remained unclear. Here, using in vivo and ex vivo experiments, we investigate the neuronal origins of altered NVC dynamics under chronic stress conditions in adult male mice. Stimulus-evoked hemodynamic and neural responses, especially beta and gamma-band local field potential activity, were significantly lower in chronically stressed animals, and the NVC relationship, itself, had changed. Further, using acute brain slices, we discovered that the underlying cause of this change was dysfunction of neuronal nitric oxide synthase (nNOS)-mediated vascular responses. Using FISH to check the mRNA expression of several GABAergic subtypes, we confirmed that only nNOS mRNA was significantly decreased in chronically stressed mice. Ultimately, chronic stress impairs NVC by diminishing nNOS-mediated vasodilation responses to local neural activity. Overall, these findings provide useful information in understanding NVC dynamics in the healthy brain. More importantly, this study reveals that impaired nNOS-mediated NVC function may be a contributory factor in the progression of stress-related diseases.SIGNIFICANCE STATEMENT The correlation between neuronal activity and cerebral vascular dynamics is defined as neurovascular coupling (NVC), which plays an important role for meeting the metabolic demands of the brain. However, the impact of chronic stress, which is a contributory factor of many cerebrovascular diseases, on NVC is poorly understood. We therefore investigated the effects of chronic stress on impaired neurovascular response to sensory stimulation and their underlying mechanisms. Multimodal approaches, from in vivo hemodynamic imaging and electrophysiology to ex vivo vascular imaging with pharmacological treatment, patch-clamp recording, FISH, and immunohistochemistry revealed that chronic stress-induced dysfunction of nNOS-expressing interneurons contributes to NVC impairment. These findings will provide useful information to understand the role of nNOS interneurons in NVC in normal and pathological conditions.
Keywords: GABAergic interneuron; cerebral blood flow; chronic stress; nNOS; neurovascular coupling.
Copyright © 2019 Han et al.
Figures





References
-
- Berwick J, Johnston D, Jones M, Martindale J, Martin C, Kennerley AJ, Redgrave P, Mayhew JE (2008) Fine detail of neurovascular coupling revealed by spatiotemporal analysis of the hemodynamic response to single whisker stimulation in rat barrel cortex. J Neurophysiol 99:787–798. 10.1152/jn.00658.2007 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
