3-Ketoacyl-ACP synthase (KAS) III homologues and their roles in natural product biosynthesis
- PMID: 31673313
- PMCID: PMC6786007
- DOI: 10.1039/c9md00162j
3-Ketoacyl-ACP synthase (KAS) III homologues and their roles in natural product biosynthesis
Abstract
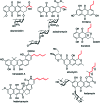
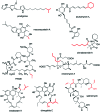
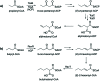
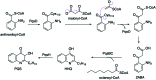
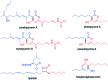
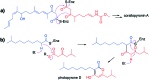
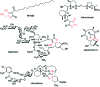
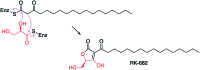
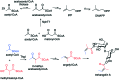
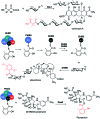
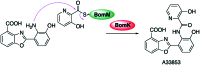
The 3-ketoacyl-ACP synthase (KAS) III proteins are one of the most abundant enzymes in nature, as they are involved in the biosynthesis of fatty acids and natural products. KAS III enzymes catalyse a carbon-carbon bond formation reaction that involves the α-carbon of a thioester and the carbonyl carbon of another thioester. In addition to the typical KAS III enzymes involved in fatty acid and polyketide biosynthesis, there are proteins homologous to KAS III enzymes that catalyse reactions that are different from that of the traditional KAS III enzymes. Those include enzymes that are responsible for a head-to-head condensation reaction, the formation of acetoacetyl-CoA in mevalonate biosynthesis, tailoring processes via C-O bond formation or esterification, as well as amide formation. This review article highlights the diverse reactions catalysed by this class of enzymes and their role in natural product biosynthesis.
This journal is © The Royal Society of Chemistry 2019.
Figures
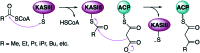






References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

